Developing Targeted Hybrid Imaging Probes by Chelator Scaffolding
- PMID: 28462989
- PMCID: PMC5481817
- DOI: 10.1021/acs.bioconjchem.7b00182
Developing Targeted Hybrid Imaging Probes by Chelator Scaffolding
Abstract
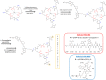
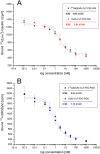
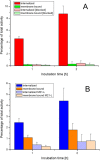
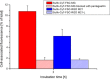
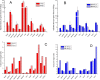
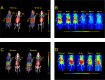
Positron emission tomography (PET) as well as optical imaging (OI) with peptide receptor targeting probes have proven their value for oncological applications but also show restrictions depending on the clinical field of interest. Therefore, the combination of both methods, particularly in a single molecule, could improve versatility in clinical routine. This proof of principle study aims to show that a chelator, Fusarinine C (FSC), can be utilized as scaffold for novel dimeric dual-modality imaging agents. Two targeting vectors (a minigastrin analogue (MG11) targeting cholecystokinin-2 receptor overexpression (CCK2R) or integrin αVβ3 targeting cyclic pentapeptides (RGD)) and a near-infrared fluorophore (Sulfo-Cyanine7) were conjugated to FSC. The probes were efficiently labeled with gallium-68 and in vitro experiments including determination of logD, stability, protein binding, cell binding, internalization, and biodistribution studies as well as in vivo micro-PET/CT and optical imaging in U-87MG αVβ3- and A431-CCK2R expressing tumor xenografted mice were carried out. Novel bioconjugates showed high receptor affinity and highly specific targeting properties at both receptors. Ex vivo biodistribution and micro-PET/CT imaging studies revealed specific tumor uptake accompanied by slow blood clearance and retention in nontargeted tissues (spleen, liver, and kidneys) leading to visualization of tumors at early (30 to 120 min p.i.). Excellent contrast in corresponding optical imaging studies was achieved especially at delayed time points (24 to 72 h p.i.). Our findings show the proof of principle of chelator scaffolding for hybrid imaging agents and demonstrate FSC being a suitable bifunctional chelator for this approach. Improvements to fine-tune pharmacokinetics are needed to translate this into a clinical setting.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

