Healthy families: study protocol for a randomized controlled trial of a screening, brief intervention, and referral to treatment intervention for caregivers to reduce secondhand smoke exposure among pediatric emergency patients
- PMID: 28464887
- PMCID: PMC5414142
- DOI: 10.1186/s12889-017-4278-8
Healthy families: study protocol for a randomized controlled trial of a screening, brief intervention, and referral to treatment intervention for caregivers to reduce secondhand smoke exposure among pediatric emergency patients
Abstract
Background: Involuntary exposure to secondhand smoke (SHSe) is an important cause of morbidity in children who present to the pediatric emergency department (PED) and urgent care (UC). SHSe interventions delivered in the PED and UC would benefit both the smoker and child, but there have been no large trials testing the efficacy of such interventions. The Healthy Families program is the first randomized controlled trial to test whether a screening, brief intervention, and referral to treatment (SBIRT) intervention delivered in the PED and UC will be effective in decreasing SHSe in children and increasing cessation in smokers.
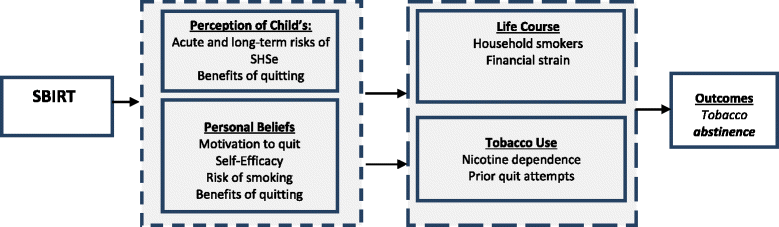
Methods/design: This trial uses a randomized, two-group design in which caregiver-smokers of children 0-17 years old are recruited from the PED and UC. Eligible caregiver-smokers are randomized to either the: 1) SBIRT Condition with face-to-face, tailored counseling that focuses on the child's illness, the importance of reducing child SHSe, caregiver smoking cessation, and the option to receive nicotine replacement therapy; or 2) Healthy Habits Control Condition which includes face-to-face, tailored attention control "5-2-1-0" counseling that focuses on improving the child's health. Dyadic assessments are conducted in-person at baseline, and via email, phone, or in-person at 6-weeks and 6-months. The primary outcomes are biochemically-verified, 7-day point prevalence and prolonged smoking abstinence. Secondary outcomes are cigarettes smoked per week, 24 h quit attempts, and biochemically validated child SHSe at each time point. The costs of this intervention will also be analyzed.
Discussion: This study will test an innovative, multilevel intervention designed to reduce child SHSe and increase smoking cessation in caregivers. If effective and routinely used, this SBIRT model could reach at least one million smokers a year in the U.S., resulting in significant reductions in caregivers' tobacco use, SHSe-related pediatric illness, and healthcare costs in this population of children.
Trial registration: ClinicalTrials.gov Identifier: NCT02531594. Date of registration: August 4, 2015.
Keywords: Child; Emergency department; Intervention; Parents/education; Pediatrics; Smoking cessation; Tobacco; Urgent care.
Figures
Similar articles
-
Kids Safe and Smokefree (KiSS): a randomized controlled trial of a multilevel intervention to reduce secondhand tobacco smoke exposure in children.BMC Public Health. 2013 Aug 30;13:792. doi: 10.1186/1471-2458-13-792. BMC Public Health. 2013. PMID: 23987302 Free PMC article. Clinical Trial.
-
Babies Living Safe & Smokefree: randomized controlled trial of a multilevel multimodal behavioral intervention to reduce low-income children's tobacco smoke exposure.BMC Public Health. 2017 Mar 14;17(1):249. doi: 10.1186/s12889-017-4145-7. BMC Public Health. 2017. PMID: 28288601 Free PMC article. Clinical Trial.
-
A smoking cessation intervention for low-income smokers in the ED.Am J Emerg Med. 2015 Aug;33(8):1056-61. doi: 10.1016/j.ajem.2015.04.058. Epub 2015 May 2. Am J Emerg Med. 2015. PMID: 25976268 Free PMC article.
-
[Smoking reduction and temporary abstinence: new approaches for smoking cessation].J Mal Vasc. 2003 Dec;28(5):293-300. J Mal Vasc. 2003. PMID: 14978435 Review. French.
-
A Mixed-Methods Systematic Review and Synthesis of Secondary Care Interventions to Reduce Secondhand Smoke Exposure Among Children and Young People.Nicotine Tob Res. 2021 Mar 19;23(4):643-653. doi: 10.1093/ntr/ntaa216. Nicotine Tob Res. 2021. PMID: 33098295
Cited by
-
Tobacco Use Behaviors and Perceptions of Parental Smokers in the Emergency Department Setting.Tob Use Insights. 2019 Jun 19;12:1179173X19841392. doi: 10.1177/1179173X19841392. eCollection 2019. Tob Use Insights. 2019. PMID: 31258335 Free PMC article.
-
Differential associations of hand nicotine and urinary cotinine with children's exposure to tobacco smoke and clinical outcomes.Environ Res. 2021 Nov;202:111722. doi: 10.1016/j.envres.2021.111722. Epub 2021 Jul 21. Environ Res. 2021. PMID: 34297932 Free PMC article.
-
Comparison of Levels of Three Tobacco Smoke Exposure Biomarkers in Children of Smokers.Int J Environ Res Public Health. 2021 Nov 10;18(22):11803. doi: 10.3390/ijerph182211803. Int J Environ Res Public Health. 2021. PMID: 34831559 Free PMC article.
-
A Parental Smoking Cessation Intervention in the Pediatric Emergency Setting: A Randomized Trial.Int J Environ Res Public Health. 2020 Nov 4;17(21):8151. doi: 10.3390/ijerph17218151. Int J Environ Res Public Health. 2020. PMID: 33158230 Free PMC article. Clinical Trial.
-
Provision of free nicotine replacement therapy to parental smokers in the pediatric emergency setting.Tob Prev Cessat. 2020 May 18;6:30. doi: 10.18332/tpc/119125. eCollection 2020. Tob Prev Cessat. 2020. PMID: 32760865 Free PMC article.
References
-
- Office on Smoking and Health (US). The Health Consequences of Involuntary Exposure to Tobacco Smoke: A Report of the Surgeon General. 2006. www.ncbi.nlm.nih.gov/books/NBK44324/. Accessed 9 Jan 2017. - PubMed
-
- U.S. Department of Health and Human Services. The Health Consequences of Smoking: 50 Years of Progress. A Report of the Surgeon General. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2014. http://www.surgeongeneral.gov/library/reports/50-years-of-progress/full-.... Accessed 9 Jan 2017.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical