Survival benefit of glioblastoma patients after FDA approval of temozolomide concomitant with radiation and bevacizumab: A population-based study
- PMID: 28467795
- PMCID: PMC5546458
- DOI: 10.18632/oncotarget.17054
Survival benefit of glioblastoma patients after FDA approval of temozolomide concomitant with radiation and bevacizumab: A population-based study
Abstract
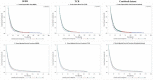
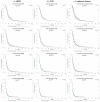
Few population-based analyses have investigated survival change in glioblastoma multiforme (GBM) patients treated with concomitant radiotherapy-temozolomide (RT-TMZ) and adjuvant temozolomide (TMZ) and then bevacizumab (BEV) after Food and Drug Administration (FDA) approval, respectively. We aimed to explore the effects on survival with RT-TMZ, adjuvant TMZ and BEV in general GBM population based on the Surveillance, Epidemiology, and End Results (SEER) and Texas Cancer Registry (TCR) databases. A total of 28933 GBM patients from SEER (N = 24578) and TCR (N = 4355) between January 2000 and December 2013 were included. Patients were grouped into three calendar periods based on date of diagnosis: pre-RT-TMZ and pre-BEV (1/2000-2/2005, P1), post-RT-TMZ and pre-BEV (3/2005-4/2009, P2), and post-RT-TMZ and post-BEV (5/2009-12/2013, P3). The association between calendar period of diagnosis and survival was analyzed in SEER and TCR, separately, by the Kaplan-Meier method and Cox proportional hazards model. We found a significant increase in median overall survival (OS) across the three periods in both populations. In multivariate models, the risk of death was significantly reduced during P2 and further decreased in P3, which remained unchanged after stratification. Comparison and validation analysis were performed in the combined dataset, and consistent results were observed. We conclude that the OS of GBM patients in a "real-world" setting has been steadily improved from January 2000 to December 2013, which likely resulted from the administrations of TMZ concomitant with RT and adjuvant TMZ for newly diagnosed GBM and then BEV for recurrent GBM after respective FDA approval.
Keywords: bevacizumab; cancer registry; glioblastoma (GBM); overall survival; temozolomide.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Lacroix M, Abi-Said D, Fourney DR, Gokaslan ZL, Shi W, DeMonte F, Lang FF, McCutcheon IE, Hassenbusch SJ, Holland E, Hess K, Michael C, Miller D, et al. A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J Neurosurg. 2001;95:190–8. - PubMed
-
- Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, Reulen HJ. ALA-Glioma Study Group. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol. 2006;7:392–401. - PubMed
-
- Bauchet L, Mathieu-Daudé H, Fabbro-Peray P, Rigau V, Fabbro M, Chinot O, Pallusseau L, Carnin C, Lainé K, Schlama A, Thiebaut A, Patru MC, Bauchet F, et al. Oncological patterns of care and outcome for 952 patients with newly diagnosed glioblastoma in 2004. Neuro Oncol. 2010;12:725–35. - PMC - PubMed
-
- Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, Bromberg JEC, Hau P, Mirimanoff RO, et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med. 2005;352:997–1003. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

