Development of an Immunochromatographic Strip Test for the Rapid Detection of Alternariol Monomethyl Ether in Fruit
- PMID: 28468234
- PMCID: PMC5450700
- DOI: 10.3390/toxins9050152
Development of an Immunochromatographic Strip Test for the Rapid Detection of Alternariol Monomethyl Ether in Fruit
Abstract
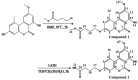
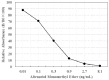
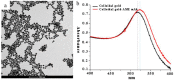
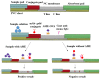
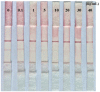
A rapid, portable, and semi-quantitative immunochromatographic strip was developed for rapid and visual detection of alternariol monomethyl ether (AME). For this purpose, the anti-AME monoclonal antibody (mAb) was prepared and identified. AME coupled to bovine serum albumin (BSA) via methyl 4-bromobutanoate was prepared as immunogen. The recoveries of AME in spiked cherry and orange fruits determined by competitive ELISA were 86.1% and 80.7%, respectively. A colloidal gold nanoparticle (CGN) and CGNs-mAb conjugate were synthesized, and on this basis, a competitive colloidal gold immunochromatographic strip was developed and applied to the detection of AME toxin in fruit samples. The intensity of red density of the test line (T line) is inversely proportional to AME concentration in the range 0.1-10 ng/mL. The visual limit of detection (LOD) of AME was found to be about 10 ng/mL. The semi-quantitative detection can be completed in 10 min. Moreover, the immunochromatographic strip has lower cross-reactivity with AME analogues, and it has a good stability performance (following 3 months of storage). Hence, the colloidal gold immunochromatographic strip could be used as a semi-quantitative tool for the on-site, rapid, and visual detection of AME in fruit.
Keywords: alternariol monomethyl ether (AME); colloidal gold nanoparticle; competitive ELISA; immunochromatographic strip test; monoclonal antibody.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
A simple, highly sensitive colorimetric immunosensor for the detection of alternariol monomethyl ether in fruit by non-aggregated gold nanoparticles.Anal Bioanal Chem. 2018 Nov;410(28):7511-7521. doi: 10.1007/s00216-018-1369-z. Epub 2018 Oct 13. Anal Bioanal Chem. 2018. PMID: 30317446
-
A microfluidic colorimetric immunoassay for sensitive detection of altenariol monomethyl ether by UV spectroscopy and smart phone imaging.Anal Chim Acta. 2019 Dec 27;1092:75-84. doi: 10.1016/j.aca.2019.09.039. Epub 2019 Sep 16. Anal Chim Acta. 2019. PMID: 31708035
-
Development of an immunochromatographic strip test for the rapid detection of zearalenone in corn.J Agric Food Chem. 2014 Nov 19;62(46):11116-21. doi: 10.1021/jf503092j. Epub 2014 Nov 10. J Agric Food Chem. 2014. PMID: 25343335
-
[Colloidal gold immunochromatographic test strip for virus detection: a review].Sheng Wu Gong Cheng Xue Bao. 2022 Sep 25;38(9):3243-3254. doi: 10.13345/j.cjb.220052. Sheng Wu Gong Cheng Xue Bao. 2022. PMID: 36151796 Review. Chinese.
-
[The comparative evaluation of components used in development of immune chromatographic test-systems].Klin Lab Diagn. 2017 Jan;62(1):30-4. Klin Lab Diagn. 2017. PMID: 30615364 Review. Russian.
Cited by
-
Simultaneous Rapid Detection of Aflatoxin B1 and Ochratoxin A in Spices Using Lateral Flow Immuno-Chromatographic Assay.Foods. 2021 Nov 9;10(11):2738. doi: 10.3390/foods10112738. Foods. 2021. PMID: 34829017 Free PMC article.
-
Assessment of the Optimum Linker Tethering Site of Alternariol Haptens for Antibody Generation and Immunoassay Development.Toxins (Basel). 2021 Dec 10;13(12):883. doi: 10.3390/toxins13120883. Toxins (Basel). 2021. PMID: 34941720 Free PMC article.
-
Recombinant Antibody-Based and Computer-Aided Comprehensive Analysis of Antibody's Equivalent Recognition Mechanism of Alternariol and Alternariol Monomethyl Ether.Front Chem. 2022 Apr 14;10:871659. doi: 10.3389/fchem.2022.871659. eCollection 2022. Front Chem. 2022. PMID: 35494638 Free PMC article.
-
A magnetic nanoparticle based immunoassay for alternariol monomethyl ether using hydrogen peroxide-mediated fluorescence quenching of CdTe quantum dots.Mikrochim Acta. 2019 Mar 7;186(4):221. doi: 10.1007/s00604-019-3334-8. Mikrochim Acta. 2019. PMID: 30847631
-
Development of Lateral Flow Immunochromatographic Assays Using Colloidal Au Sphere and Nanorods as Signal Marker for the Determination of Zearalenone in Cereals.Foods. 2020 Mar 4;9(3):281. doi: 10.3390/foods9030281. Foods. 2020. PMID: 32143348 Free PMC article.
References
-
- Alexander J., Benford D., Boobis A., Ceccatelli S., Cottrill B., Cravedi J., Di Domenico A., Doerge D., Dogliotti E., Edler L. Scientific opinion on the risks for animal and public health related to the presence of Alternaria toxins in feed and food. EFSA J. 2011;9:2407–2504.
-
- Man Y., Liang G., Li A., Pan L. Analytical methods for the determination of Alternaria mycotoxins. Chromatographia. 2016;80:1–14. doi: 10.1007/s10337-016-3186-x. - DOI
-
- Liu G.T., Qian Y.Z., Zhang P., Dong W.H., Qi Y.M., Guo H.T. Etiological role of Alternaria alternata in human esophageal cancer. Chin. Med. J. 1992;105:394–400. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

