Ube3a loss increases excitability and blunts orientation tuning in the visual cortex of Angelman syndrome model mice
- PMID: 28468997
- PMCID: PMC5511875
- DOI: 10.1152/jn.00618.2016
Ube3a loss increases excitability and blunts orientation tuning in the visual cortex of Angelman syndrome model mice
Abstract
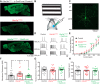
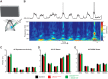
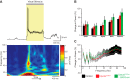
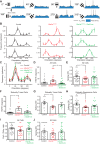
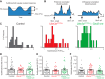
Angelman syndrome (AS) is a neurodevelopmental disorder caused by loss of the maternally inherited allele of UBE3AUbe3aSTOP/p+ mice recapitulate major features of AS in humans and allow conditional reinstatement of maternal Ube3a with the expression of Cre recombinase. We have recently shown that AS model mice exhibit reduced inhibitory drive onto layer (L)2/3 pyramidal neurons of visual cortex, which contributes to a synaptic excitatory/inhibitory imbalance. However, it remains unclear how this loss of inhibitory drive affects neural circuits in vivo. Here we examined visual cortical response properties in individual neurons to explore the consequences of Ube3a loss on intact cortical circuits and processing. Using in vivo patch-clamp electrophysiology, we measured the visually evoked responses to square-wave drifting gratings in L2/3 regular-spiking (RS) neurons in control mice, Ube3a-deficient mice, and mice in which Ube3a was conditionally reinstated in GABAergic neurons. We found that Ube3a-deficient mice exhibited enhanced pyramidal neuron excitability in vivo as well as weaker orientation tuning. These observations are the first to show alterations in cortical computation in an AS model, and they suggest a basis for cortical dysfunction in AS.NEW & NOTEWORTHY Angelman syndrome (AS) is a severe neurodevelopmental disorder caused by the loss of the gene UBE3A Using electrophysiological recording in vivo, we describe visual cortical dysfunctions in a mouse model of AS. Aberrant cellular properties in AS model mice could be improved by reinstating Ube3a in inhibitory neurons. These findings suggest that inhibitory neurons play a substantial role in the pathogenesis of AS.
Keywords: Angelman syndrome; Ube3a; autism; visual cortex.
Copyright © 2017 the American Physiological Society.
Figures


References
-
- Albrecht DG, Hamilton DB. Striate cortex of monkey and cat: contrast response function. J Neurophysiol 48: 217–237, 1982. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

