Investigation of cationicity and structure of pseudin-2 analogues for enhanced bacterial selectivity and anti-inflammatory activity
- PMID: 28469145
- PMCID: PMC5431190
- DOI: 10.1038/s41598-017-01474-0
Investigation of cationicity and structure of pseudin-2 analogues for enhanced bacterial selectivity and anti-inflammatory activity
Abstract
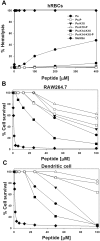
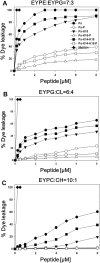
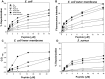
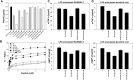
Pseudin-2 (Ps), isolated from the frog Pseudis paradoxa, exhibits potent antibacterial activity and cytotoxicity. To develop antimicrobial peptides with anti-inflammatory activity and low cytotoxicity, we designed Ps analogues with Lys substitutions, resulting in elevated amphipathic α-helical structure and cationicity. We further substituted Gly11 with Pro (Ps-P analogues) to increase bacterial cell selectivity. Ps analogues retained antimicrobial activity and exhibited reduced cytotoxicity, whereas Ps-P analogues exhibited lower cytotoxicity and antimicrobial activity. Tertiary structures revealed that Ps has a linear α-helix from Leu2 to Glu24, whereas Ps-P has a bend at Pro11 between two short α-helixes. Using various biophysical experiments, we found that Ps analogues produced much higher membrane depolarization than Ps-P analogues, whereas Ps-P analogues may penetrate bacterial cell membranes. Ps and its analogue Ps-K18 exhibited potent anti-inflammatory activity in LPS-stimulated RAW264.7 and mouse dendritic cells via a mechanism involving the Toll-like receptor 4 (TLR4) pathway. These activities may arise from their direct inhibition of the formation of TLR4-MD-2_LPS complex, implying that amphipathic α-helical structure with an optimum balance between enhanced cationicity and hydrophobicity may be essential for their anti-inflammatory activity. The bent structure provided by Pro substitution plays an important role in enhancing bacterial cell selectivity and cell penetration.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
Pse-T2, an Antimicrobial Peptide with High-Level, Broad-Spectrum Antimicrobial Potency and Skin Biocompatibility against Multidrug-Resistant Pseudomonas aeruginosa Infection.Antimicrob Agents Chemother. 2018 Nov 26;62(12):e01493-18. doi: 10.1128/AAC.01493-18. Print 2018 Dec. Antimicrob Agents Chemother. 2018. PMID: 30323036 Free PMC article.
-
Design of potent, non-toxic antimicrobial agents based upon the structure of the frog skin peptide, pseudin-2.Regul Pept. 2005 Jul 15;129(1-3):85-91. doi: 10.1016/j.regpep.2005.01.015. Regul Pept. 2005. PMID: 15927702
-
Effects of Aib residues insertion on the structural-functional properties of the frog skin-derived peptide esculentin-1a(1-21)NH2.Amino Acids. 2017 Jan;49(1):139-150. doi: 10.1007/s00726-016-2341-x. Epub 2016 Oct 11. Amino Acids. 2017. PMID: 27726008
-
An intimate link between antimicrobial peptide sequence diversity and binding to essential components of bacterial membranes.Biochim Biophys Acta. 2016 May;1858(5):958-70. doi: 10.1016/j.bbamem.2015.10.011. Epub 2015 Oct 21. Biochim Biophys Acta. 2016. PMID: 26498397 Review.
-
Recent Advances in the Exploration of Therapeutic Analogues of Gramicidin S, an Old but Still Potent Antimicrobial Peptide.J Med Chem. 2019 Sep 12;62(17):7603-7617. doi: 10.1021/acs.jmedchem.9b00156. Epub 2019 Apr 17. J Med Chem. 2019. PMID: 30938996 Review.
Cited by
-
Antiseptic Effect of Ps-K18: Mechanism of Its Antibacterial and Anti-Inflammatory Activities.Int J Mol Sci. 2019 Oct 2;20(19):4895. doi: 10.3390/ijms20194895. Int J Mol Sci. 2019. PMID: 31581682 Free PMC article.
-
Pse-T2, an Antimicrobial Peptide with High-Level, Broad-Spectrum Antimicrobial Potency and Skin Biocompatibility against Multidrug-Resistant Pseudomonas aeruginosa Infection.Antimicrob Agents Chemother. 2018 Nov 26;62(12):e01493-18. doi: 10.1128/AAC.01493-18. Print 2018 Dec. Antimicrob Agents Chemother. 2018. PMID: 30323036 Free PMC article.
-
Target Proteins of Phloretin for Its Anti-Inflammatory and Antibacterial Activities Against Propionibacterium acnes-Induced Skin Infection.Molecules. 2019 Apr 3;24(7):1319. doi: 10.3390/molecules24071319. Molecules. 2019. PMID: 30987239 Free PMC article.
-
Phloretin as a Potent Natural TLR2/1 Inhibitor Suppresses TLR2-Induced Inflammation.Nutrients. 2018 Jul 5;10(7):868. doi: 10.3390/nu10070868. Nutrients. 2018. PMID: 29976865 Free PMC article.
-
Anti-Endotoxin 9-Meric Peptide with Therapeutic Potential for the Treatment of Endotoxemia.J Microbiol Biotechnol. 2021 Jan 28;31(1):25-32. doi: 10.4014/jmb.2011.11011. J Microbiol Biotechnol. 2021. PMID: 33263333 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

