Simultaneous Quantification of Amino Metabolites in Multiple Metabolic Pathways Using Ultra-High Performance Liquid Chromatography with Tandem-mass Spectrometry
- PMID: 28469184
- PMCID: PMC5431165
- DOI: 10.1038/s41598-017-01435-7
Simultaneous Quantification of Amino Metabolites in Multiple Metabolic Pathways Using Ultra-High Performance Liquid Chromatography with Tandem-mass Spectrometry
Abstract
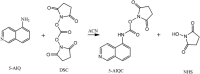
Metabolites containing amino groups cover multiple pathways and play important roles in redox homeostasis and biosyntheses of proteins, nucleotides and neurotransmitters. Here, we report a new method for simultaneous quantification of 124 such metabolites. This is achieved by derivatization-assisted sensitivity enhancement with 5-aminoisoquinolyl-N-hydroxysuccinimidyl carbamate (5-AIQC) followed with comprehensive analysis using ultra-high performance liquid chromatography and electrospray ionization tandem mass spectrometry (UHPLC-MS/MS). In an one-pot manner, this quantification method enables simultaneous coverage of 20 important metabolic pathways including protein biosynthesis/degradation, biosyntheses of catecholamines, arginine and glutathione, metabolisms of homocysteine, taurine-hypotaurine etc. Compared with the reported ones, this method is capable of simultaneously quantifying thiols, disulfides and other oxidation-prone analytes in a single run and suitable for quantifying aromatic amino metabolites. This method is also much more sensitive for all tested metabolites with LODs well below 50 fmol (at sub-fmol for most tested analytes) and shows good precision for retention time and quantitation with inter-day and intra-day relative standard deviations (RSDs) below 15% and good recovery from renal cancer tissue, rat urine and plasma. The method was further applied to quantify the amino metabolites in silkworm hemolymph from multiple developmental stages showing its applicability in metabolomics and perhaps some clinical chemistry studies.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
A strategy for the targeted metabolomics analysis of 11 gut microbiota-host co-metabolites in rat serum, urine and feces by ultra high performance liquid chromatography-tandem mass spectrometry.J Chromatogr A. 2016 Jan 15;1429:207-17. doi: 10.1016/j.chroma.2015.12.031. Epub 2015 Dec 14. J Chromatogr A. 2016. PMID: 26733392
-
Determination of dopamine, serotonin, biosynthesis precursors and metabolites in rat brain microdialysates by ultrasonic-assisted in situ derivatization-dispersive liquid-liquid microextraction coupled with UHPLC-MS/MS.Talanta. 2016 Dec 1;161:253-264. doi: 10.1016/j.talanta.2016.08.036. Epub 2016 Aug 16. Talanta. 2016. PMID: 27769404
-
A new combined method of stable isotope-labeling derivatization-ultrasound-assisted dispersive liquid-liquid microextraction for the determination of neurotransmitters in rat brain microdialysates by ultra high performance liquid chromatography tandem mass spectrometry.J Chromatogr B Analyt Technol Biomed Life Sci. 2017 Jun 1;1054:64-72. doi: 10.1016/j.jchromb.2017.03.039. Epub 2017 Apr 10. J Chromatogr B Analyt Technol Biomed Life Sci. 2017. PMID: 28426993
-
In situ derivatization-ultrasound-assisted dispersive liquid-liquid microextraction for the determination of neurotransmitters in Parkinson's rat brain microdialysates by ultra high performance liquid chromatography-tandem mass spectrometry.J Chromatogr A. 2016 Aug 5;1458:70-81. doi: 10.1016/j.chroma.2016.06.059. Epub 2016 Jun 18. J Chromatogr A. 2016. PMID: 27372412
-
Isotope-coded ESI-enhancing derivatization reagents for differential analysis, quantification and profiling of metabolites in biological samples by LC/MS: A review.J Pharm Biomed Anal. 2016 Oct 25;130:181-193. doi: 10.1016/j.jpba.2016.04.033. Epub 2016 Apr 30. J Pharm Biomed Anal. 2016. PMID: 27178301 Review.
Cited by
-
Effects of replacing fishmeal with cottonseed protein concentrate on growth performance, blood metabolites, and the intestinal health of juvenile rainbow trout (Oncorhynchus mykiss).Front Immunol. 2022 Dec 21;13:1079677. doi: 10.3389/fimmu.2022.1079677. eCollection 2022. Front Immunol. 2022. PMID: 36618404 Free PMC article.
-
Challenges for cysteamine stabilization, quantification, and biological effects improvement.J Pharm Anal. 2020 Dec;10(6):499-516. doi: 10.1016/j.jpha.2020.03.007. Epub 2020 Mar 21. J Pharm Anal. 2020. PMID: 33425447 Free PMC article. Review.
-
SARS-CoV-2 and HCoV-OC43 regulate host m6A modification via activation of the mTORC1 signalling pathway to facilitate viral replication.Emerg Microbes Infect. 2025 Dec;14(1):2447620. doi: 10.1080/22221751.2024.2447620. Epub 2025 Feb 24. Emerg Microbes Infect. 2025. PMID: 39745173 Free PMC article.
-
Translational Metabolomics: Current Challenges and Future Opportunities.Metabolites. 2019 Jun 6;9(6):108. doi: 10.3390/metabo9060108. Metabolites. 2019. PMID: 31174372 Free PMC article.
-
Challenges in Metabolomics-Based Tests, Biomarkers Revealed by Metabolomic Analysis, and the Promise of the Application of Metabolomics in Precision Medicine.Int J Mol Sci. 2022 May 6;23(9):5213. doi: 10.3390/ijms23095213. Int J Mol Sci. 2022. PMID: 35563604 Free PMC article. Review.
References
-
- Tang H, Wang Y. Metabonomics: a revolution in progress. Progress in Biochemistry and Biophysics. 2005;33:401–417.
-
- Fan TW-M. Metabolite profiling by one-and two-dimensional NMR analysis of complex mixtures. Progress in nuclear magnetic resonance spectroscopy. 1996;28:161–219. doi: 10.1016/0079-6565(96)90002-3. - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

