Targeted apoptosis in ovarian cancer cells through mitochondrial dysfunction in response to Sambucus nigra agglutinin
- PMID: 28471452
- PMCID: PMC5520748
- DOI: 10.1038/cddis.2017.77
Targeted apoptosis in ovarian cancer cells through mitochondrial dysfunction in response to Sambucus nigra agglutinin
Abstract
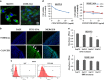
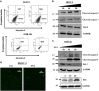
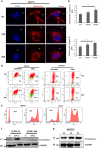
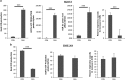
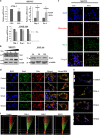
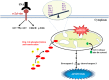
Ovarian carcinoma (OC) patients encounter the severe challenge of clinical management owing to lack of screening measures, chemoresistance and finally dearth of non-toxic therapeutics. Cancer cells deploy various defense strategies to sustain the tumor microenvironment, among which deregulated apoptosis remains a versatile promoter of cancer progression. Although recent research has focused on identifying agents capable of inducing apoptosis in cancer cells, yet molecules efficiently breaching their survival advantage are yet to be classified. Here we identify lectin, Sambucus nigra agglutinin (SNA) to exhibit selectivity towards identifying OC by virtue of its specific recognition of α-2, 6-linked sialic acids. Superficial binding of SNA to the OC cells confirm the hyper-sialylated status of the disease. Further, SNA activates the signaling pathways of AKT and ERK1/2, which eventually promotes de-phosphorylation of dynamin-related protein-1 (Drp-1). Upon its translocation to the mitochondrial fission loci Drp-1 mediates the central role of switch in the mitochondrial phenotype to attain fragmented morphology. We confirmed mitochondrial outer membrane permeabilization resulting in ROS generation and cytochrome-c release into the cytosol. SNA response resulted in an allied shift of the bioenergetics profile from Warburg phenotype to elevated mitochondrial oxidative phosphorylation, altogether highlighting the involvement of mitochondrial dysfunction in restraining cancer progression. Inability to replenish the SNA-induced energy crunch of the proliferating cancer cells on the event of perturbed respiratory outcome resulted in cell cycle arrest before G2/M phase. Our findings position SNA at a crucial juncture where it proves to be a promising candidate for impeding progression of OC. Altogether we unveil the novel aspect of identifying natural molecules harboring the inherent capability of targeting mitochondrial structural dynamics, to hold the future for developing non-toxic therapeutics for treating OC.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

