STAT2 is involved in the pathogenesis of psoriasis by promoting CXCL11 and CCL5 production by keratinocytes
- PMID: 28472186
- PMCID: PMC5417613
- DOI: 10.1371/journal.pone.0176994
STAT2 is involved in the pathogenesis of psoriasis by promoting CXCL11 and CCL5 production by keratinocytes
Abstract
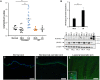
The JAK/STAT signaling pathway is suggested to play an important role in the pathogenesis of psoriasis, and recently JAK/STAT inhibitors have shown promising results in psoriasis treatment. The present study aimed to characterize the role of STAT2 in psoriasis. We demonstrated an increased expression of STAT2 and an increased level of phosphorylated/activated STAT2 in lesional compared with nonlesional psoriatic skin. Gene silencing of STAT2 by siRNA in human keratinocytes revealed that upon IFNα stimulation CXCL11 and CCL5 were the only two cytokines, among 102 analyzed, found to be regulated through a STAT2-dependent mechanism. Moreover, the regulation of CXCL11 and CCL5 depended on IRF9, but not on STAT1 and STAT6. The CXCL11 and CCL5 expression was increased in lesional compared with nonlesional psoriatic skin, and analysis demonstrated positive correlation between the expression of CXCL11 and IFNγ and between the expression of CCL5 and IFNγ in lesional psoriatic skin. In contrast, no correlation between the expression of CXCL11 and IL-17A and the expression of CCL5 and IL-17A in lesional psoriatic skin was found. Our data suggest that STAT2 plays a role in the psoriasis pathogenesis by regulating the expression of CXCL11 and CCL5, and thereby attracting IFNγ-producing immune cells to the skin.
Conflict of interest statement
Figures

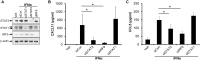


References
-
- Nickoloff BJ, Qin JZ, Nestle FO. Immunopathogenesis of psoriasis. Clinical reviews in allergy & immunology. 2007;33(1–2):45–56. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

