Allosteric Modulation of GPCRs: New Insights and Potential Utility for Treatment of Schizophrenia and Other CNS Disorders
- PMID: 28472649
- PMCID: PMC5482176
- DOI: 10.1016/j.neuron.2017.03.016
Allosteric Modulation of GPCRs: New Insights and Potential Utility for Treatment of Schizophrenia and Other CNS Disorders
Abstract
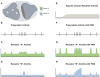
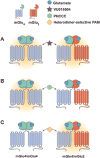
G-protein-coupled receptors (GPCRs) play critical roles in regulating brain function. Recent advances have greatly expanded our understanding of these receptors as complex signaling machines that can adopt numerous conformations and modulate multiple downstream signaling pathways. While agonists and antagonists have traditionally been pursued to target GPCRs, allosteric modulators provide several mechanistic advantages, including the ability to distinguish between closely related receptor subtypes. Recently, the discovery of allosteric ligands that confer bias and modulate some, but not all, of a given receptor's downstream signaling pathways can provide pharmacological modulation of brain circuitry with remarkable precision. In addition, allosteric modulators with unprecedented specificity have been developed that can differentiate between subpopulations of a given receptor subtype based on the receptor's dimerization state. These advances are not only providing insight into the biological roles of specific receptor populations, but hold great promise for treating numerous CNS disorders.
Keywords: GPCR; NAM; PAM; allosteric modulator; dopamine receptor; mAChR; mGlu; metabotropic glutamate receptor; muscarinic receptor; schizophrenia.
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures


References
-
- Ade KK, Wan Y, Hamann HC, O'Hare JK, Guo W, Quian A, Kumar S, Bhagat S, Rodriguiz RM, Wetsel WC, et al. Increased Metabotropic Glutamate Receptor 5 Signaling Underlies Obsessive-Compulsive Disorder-like Behavioral and Striatal Circuit Abnormalities in Mice. Biol Psychiatry. 2016;80:522–533. - PMC - PubMed
-
- Aiba A, Chen C, Herrup K, Rosenmund C, Stevens CF, Tonegawa S. Reduced hippocampal long-term potentiation and context-specific deficit in associative learning in mGluR1 mutant mice. Cell. 1994;79:365–375. - PubMed
-
- Airan RD, Thompson KR, Fenno LE, Bernstein H, Deisseroth K. Temporally precise in vivo control of intracellular signalling. Nature. 2009;458:1025–1029. - PubMed
-
- Alt A, Pendri A, Bertekap RL, Jr, Li G, Benitex Y, Nophsker M, Rockwell KL, Burford NT, Sum CS, Chen J, et al. Evidence for Classical Cholinergic Toxicity Associated with Selective Activation of M1 Muscarinic Receptors. J Pharmacol Exp Ther. 2016;356:293–304. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

