Efficacy and safety of gemcitabine plus S-1 in pancreatic cancer: a pooled analysis of individual patient data
- PMID: 28472821
- PMCID: PMC5518857
- DOI: 10.1038/bjc.2017.128
Efficacy and safety of gemcitabine plus S-1 in pancreatic cancer: a pooled analysis of individual patient data
Abstract
Background: Three randomised trials (GEST, JACCRO PC-01, and GEMSAP) were conducted to evaluate the efficacy of gemcitabine plus S-1 (GS) vs gemcitabine alone in patients with advanced pancreatic cancer (PC). In this pooled analysis, the efficacy and safety of GS vs gemcitabine were evaluated.
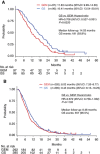
Methods: Additional follow-up was conducted and survival data were updated in each study. A total of 770 patients (gemcitabine 389; GS 381) were included in the pooled analysis. The efficacy and safety data were analysed according to disease extent: locally advanced PC (LAPC) or metastatic PC (MPC).
Results: There were 738 (95.8%) overall survival events. In patients with LAPC (n=193), the median survival was 11.83 months for gemcitabine and 16.41 months for GS (hazard ratio (HR)=0.708; 95% confidence intervals (CI), 0.527-0.951; P=0.0220). In patients with MPC (n=577), the median survival was 8.02 months for gemcitabine and 9.43 months for GS (HR=0.872; 95% CI, 0.738-1.032; P=0.1102). The rate of grade 3/4 toxicity (rash and thrombocytopenia in LAPC; rash, diarrhoea, vomiting, and neutropaenia in MPC) was significantly higher for GS than for gemcitabine.
Conclusions: Gemcitabine plus S-1 is a viable treatment alternative to gemcitabine, which is one of the standard treatments in patients with LAPC.
Conflict of interest statement
CH has received consultancies from Taiho Pharmaceutical Co., Ltd. TO has received honoraria from Taiho Pharmaceutical Co., Ltd., Eli Lilly Japan KK, consultancies and research funding from Eli Lilly Japan KK and Taiho Pharmaceutical Co., Ltd. H Isayama has received honoraria from Taiho Pharmaceutical Co., Ltd. and Eli Lilly Japan KK, and research funding from Taiho Pharmaceutical Co., Ltd. JF has received honoraria from Taiho Pharmaceutical Co., Ltd. and Eli Lilly Japan KK, consultancies from Taiho Pharmaceutical Co., Ltd., and research funding from Taiho Pharmaceutical Co., Ltd. and Eli Lilly Japan KK H Ishii, has received honoraria from Taiho Pharmaceutical Co., Ltd. and Eli Lilly Japan KK, and research funding from Taiho Pharmaceutical Co., Ltd. The remaining authors declare no conflict of interest.
Figures
References
-
- Berlin JD, Catalano P, Thomas JP, Kugler JW, Haller DG, Benson AB 3rd (2002) Phase III study of gemcitabine in combination with fluorouracil versus gemcitabine alone in patients with advanced pancreatic carcinoma: Eastern Cooperative Oncology Group Trial E2297. J Clin Oncol 20: 3270–3275. - PubMed
-
- Burris HA 3rd, Moore MJ, Andersen J, Green MR, Rothenberg ML, Modiano MR, Cripps MC, Portenoy RK, Storniolo AM, Tarassoff P, Nelson R, Dorr FA, Stephens CD, Von Hoff DD (1997) Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 15: 2403–2413. - PubMed
-
- Cherny NI, Sullivan R, Dafni U, Kerst JM, Sobrero A, Zielinski C, de Vries EG, Piccart MJ (2015) A standardised, generic, validated approach to stratify the magnitude of clinical benefit that can be anticipated from anti-cancer therapies: the European Society for Medical Oncology Magnitude of Clinical Benefit Scale (ESMO-MCBS). Ann Oncol 26(8): 1547–1573. - PubMed
-
- Chuah B, Goh BC, Lee SC, Soong R, Lau F, Mulay M, Dinolfo M, Lim SE, Soo R, Furuie T, Saito K, Zergebel C, Rosen LS (2011) Comparison of the pharmacokinetics and pharmacodynamics of S-1 between European and East Asian patients. Cancer Sci 102(2): 478–483. - PubMed
-
- Conroy T, Desseigne F, Ychou M, Bouché O, Guimbaud R, Bécouarn Y, Adenis A, Raoul JL, Gourgou-Bourgade S, de la Fouchardière C, Bennouna J, Bachet JB, Khemissa-Akouz F, Péré-Vergé D, Delbaldo C, Assenat E, Chauffert B, Michel P, Montoto-Grillot C, Ducreux M Groupe Tumeurs Digestives of Unicancer; PRODIGE Intergroup (2011) FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 364: 1817–1825. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical