A QA Program for MRD Testing Demonstrates That Systematic Education Can Reduce Discordance Among Experienced Interpreters
- PMID: 28475275
- PMCID: PMC8064036
- DOI: 10.1002/cyto.b.21528
A QA Program for MRD Testing Demonstrates That Systematic Education Can Reduce Discordance Among Experienced Interpreters
Abstract
Background: Minimal residual disease (MRD) in B lymphoblastic leukemia (B-ALL) by flow cytometry is an established prognostic factor used to adjust treatment in most pediatric therapeutic protocols. MRD in B-ALL has been standardized by the Children's Oncology Group (COG) in North America, but not routine clinical labs. The Foundation for National Institutes of Health sought to harmonize MRD measurement among COG, oncology groups, academic, community and government, laboratories.
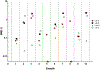
Methods: Listmode data from post-induction marrows were distributed from a reference lab to seven different clinical FCM labs with variable experience in B-ALL MRD. Labs were provided with the COG protocol. Files from 15 cases were distributed to the seven labs. Educational sessions were implemented, and 10 more listmode file cases analyzed.
Results: Among 105 initial challenges, the overall discordance rate was 26%. In the final round, performance improved considerably; out of 70 challenges, there were five false positives and one false negative (9% discordance), and no quantitative discordance. Four of six deviations occurred in a single lab. Three samples with hematogones were still misclassified as MRD.
Conclusions: Despite the provision of the COG standardized analysis protocol, even experienced laboratories require an educational component for B-ALL MRD analysis by FCM. Recognition of hematogones remains challenging for some labs when using the COG protocol. The results from this study suggest that dissemination of MRD testing to other North American laboratories as part of routine clinical management of B-ALL is possible but requires additional educational components to complement standardized methodology. © 2017 International Clinical Cytometry Society.
Keywords: minimal residual disease.
© 2017 International Clinical Cytometry Society.
Figures



References
-
- van Dongen JJ, Seriu T, Panzer-Grumayer ER, Biondi A, Pongers-Willemse MJ, Corral L, Stolz F, Schrappe M, Masera G, Kamps WA, et al. Prognostic value of minimal residual disease in acute lymphoblastic leukaemia in childhood. Lancet 1998;352:1731–1738. - PubMed
-
- Borowitz MJ, Devidas M, Hunger SP, Bowman WP, Carroll AJ, Carroll WL, Linda S, Martin PL, Pullen DJ, Viswanatha D, et al. Clinical significance of minimal residual disease in childhood acute lymphoblastic leukemia and its relationship to other prognostic factors: A Children’s Oncology Group study. Blood 2008;111:5477–5485. - PMC - PubMed
-
- Paiva B, Vidriales M-B, Cervero J, Mateo G, Perez JJ, Montalban MA, Sureda A, Montejano L, Gutierrez NC, Garcia de Coca A, et al. Multiparameter flow cytometric remission is the most relevant prognostic factor for multiple myeloma patients who undergo autologous stem cell transplantation. Blood 2008;112:4017–4023. - PMC - PubMed
-
- Buccisano F, Maurillo L, Del Principe MI, Del Poeta G, Sconocchia G, Lo-Coco F, Arcese W, Amadori S, Venditti A. Prognostic and therapeutic implications of minimal residual disease detection in acute myeloid leukemia. Blood 2012;119:332–341. - PubMed
-
- Paiva B, Cedena M-T, Puig N, Arana P, Vidriales M-B, Cordon L, Flores-Montero J, Gutierrez NC, Martin-Ramos M-L, Martinez-Lopez J, et al. Minimal residual disease monitoring and immune profiling in multiple myeloma in elderly patients. Blood 2016;127:3165–3174. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

