REEP6 deficiency leads to retinal degeneration through disruption of ER homeostasis and protein trafficking
- PMID: 28475715
- PMCID: PMC5808736
- DOI: 10.1093/hmg/ddx149
REEP6 deficiency leads to retinal degeneration through disruption of ER homeostasis and protein trafficking
Abstract
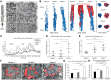
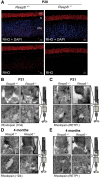
Retinitis pigmentosa (RP) is the most common form of inherited retinal dystrophy. We recently identified mutations in REEP6, which encodes the receptor expression enhancing protein 6, in several families with autosomal recessive RP. REEP6 is related to the REEP and Yop1p family of ER shaping proteins and potential receptor accessory proteins, but the role of REEP6 in the retina is unknown. Here we characterize the disease mechanisms associated with loss of REEP6 function using a Reep6 knockout mouse generated by CRISPR/Cas9 gene editing. In control mice REEP6 was localized to the inner segment and outer plexiform layer of rod photoreceptors. The Reep6-/- mice exhibited progressive photoreceptor degeneration from P20 onwards. Ultrastructural analyses at P20 by transmission electron microscopy and 3View serial block face scanning EM revealed an expansion of the distal ER in the Reep6-/- rods and an increase in their number of mitochondria. Electroretinograms revealed photoreceptor dysfunction preceded degeneration, suggesting potential defects in phototransduction. There was no effect on the traffic of rhodopsin, Rom1 or peripherin/rds; however, the retinal guanylate cyclases GC1 and GC2 were severely affected in the Reep6 knockout animals, with almost undetectable expression. These changes correlated with an increase in C/EBP homologous protein (CHOP) expression and the activation of caspase 12, suggesting that ER stress contributes to cell death. Collectively, these data suggest that REEP6 plays an essential role in maintaining cGMP homeostasis though facilitating the stability and/or trafficking of guanylate cyclases and maintaining ER and mitochondrial homeostasis.
© The Author 2017. Published by Oxford University Press.
Figures






References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

