Autocrine activation of the IFN signaling pathway may promote immune escape in glioblastoma
- PMID: 28475775
- PMCID: PMC5596176
- DOI: 10.1093/neuonc/nox051
Autocrine activation of the IFN signaling pathway may promote immune escape in glioblastoma
Abstract
Background: Interferons (IFNs) are cytokines typically induced upon viral infection but are constitutively expressed also in the absence of acute infection. The physiological role of autocrine and paracrine IFN signaling, however, remains poorly understood, and its function in glioblastoma has not been explored in depth.
Methods: Using RNA interference-mediated gene silencing, we characterized constitutive type I IFN signaling and its role in human glioma cells.
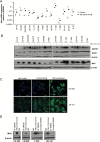
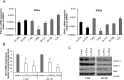
Results: We observed constitutive expression of phosphorylated signal transducer and activator of transcription 1 (pSTAT1) and myxovirus resistance protein A (MxA), a classical IFN-response marker, in the absence of exogenous IFN-β. In vivo, we found higher MxA expression in gliomas than in normal tissue, suggesting that IFN signaling is constitutively active in these tumors. To demonstrate the presence of an autocrine type I IFN signaling loop in glioma cells in vitro, we first confirmed the expression of the type I alpha/beta receptor (IFNAR)1/2, and its ligands, IFN-α and IFN-β. Small interfering RNA-mediated receptor gene silencing resulted in reduced expression of MxA at mRNA and protein levels, as did gene silencing of the ligands, corroborating the hypothesis of an autocrine signaling loop in which type I IFNs induce intracellular signaling through IFNAR1/2. On a functional level, following IFNAR1 or IFNAR2 gene silencing, we observed reduced programmed death ligand 1 (PD-L1) and major histocompatibility complex (MHC) class I and II expression as well as an enhanced susceptibility to natural killer immune cell lysis, suggesting that autocrine IFN signaling contributes to the immune evasion of glioma cells.
Conclusions: Our findings point to an important role of constitutive IFN signaling in glioma cells by modulating their interaction with the microenvironment.
Keywords: PD-L1; STAT1; glioma; immunogenicity; interferon.
© The Author(s) 2017. Published by Oxford University Press on behalf of the Society for Neuro-Oncology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com
Figures



Similar articles
-
IFNAR1 Controls Autocrine Type I IFN Regulation of PD-L1 Expression in Myeloid-Derived Suppressor Cells.J Immunol. 2018 Jul 1;201(1):264-277. doi: 10.4049/jimmunol.1800129. Epub 2018 May 11. J Immunol. 2018. PMID: 29752314 Free PMC article.
-
IL-4 suppresses dendritic cell response to type I interferons.J Immunol. 2007 Nov 15;179(10):6446-55. doi: 10.4049/jimmunol.179.10.6446. J Immunol. 2007. PMID: 17982033
-
Luteolin sensitizes the antiproliferative effect of interferon α/β by activation of Janus kinase/signal transducer and activator of transcription pathway signaling through protein kinase A-mediated inhibition of protein tyrosine phosphatase SHP-2 in cancer cells.Cell Signal. 2014 Mar;26(3):619-28. doi: 10.1016/j.cellsig.2013.11.039. Epub 2013 Dec 12. Cell Signal. 2014. PMID: 24333668
-
The molecular basis for differential type I interferon signaling.J Biol Chem. 2017 May 5;292(18):7285-7294. doi: 10.1074/jbc.R116.774562. Epub 2017 Mar 13. J Biol Chem. 2017. PMID: 28289098 Free PMC article. Review.
-
Type I interferon: a new player in TNF signaling.Curr Dir Autoimmun. 2010;11:94-104. doi: 10.1159/000289199. Epub 2010 Feb 18. Curr Dir Autoimmun. 2010. PMID: 20173389 Free PMC article. Review.
Cited by
-
Allicin Inhibits Proliferation by Decreasing IL-6 and IFN-β in HCMV-Infected Glioma Cells.Cancer Manag Res. 2020 Aug 17;12:7305-7317. doi: 10.2147/CMAR.S259677. eCollection 2020. Cancer Manag Res. 2020. PMID: 32884345 Free PMC article.
-
Glioma stem-like cells evade interferon suppression through MBD3/NuRD complex-mediated STAT1 downregulation.J Exp Med. 2020 May 4;217(5):e20191340. doi: 10.1084/jem.20191340. J Exp Med. 2020. PMID: 32181805 Free PMC article.
-
Type I IFNs contribute to upregulation of PD-L1 during Chlamydia trachomatis infection.Infect Immun. 2025 Apr 8;93(4):e0004025. doi: 10.1128/iai.00040-25. Epub 2025 Mar 12. Infect Immun. 2025. PMID: 40071913 Free PMC article.
-
Functional analysis of the short splicing variant encoded by CHI3L1/YKL-40 in glioblastoma.Front Oncol. 2022 Nov 2;12:910728. doi: 10.3389/fonc.2022.910728. eCollection 2022. Front Oncol. 2022. PMID: 36408158 Free PMC article.
-
Virus-Based Immunotherapy of Glioblastoma.Cancers (Basel). 2019 Feb 5;11(2):186. doi: 10.3390/cancers11020186. Cancers (Basel). 2019. PMID: 30764570 Free PMC article. Review.
References
-
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–674. - PubMed
-
- Weller M, Wick W, Aldape K, et al. Glioma. Nat Rev Dis Primers. 2015;1:15017. - PubMed
-
- Essers MA, Offner S, Blanco-Bose WE, et al. IFNalpha activates dormant haematopoietic stem cells in vivo. Nature. 2009;458(7240):904–908. - PubMed
-
- Sato T, Onai N, Yoshihara H, Arai F, Suda T, Ohteki T. Interferon regulatory factor-2 protects quiescent hematopoietic stem cells from type I interferon-dependent exhaustion. Nat Med. 2009;15(6):696–700. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

