Ablation of Hepatic Production of the Acid-Labile Subunit in Bovine-GH Transgenic Mice: Effects on Organ and Skeletal Growth
- PMID: 28475811
- PMCID: PMC5551555
- DOI: 10.1210/en.2016-1952
Ablation of Hepatic Production of the Acid-Labile Subunit in Bovine-GH Transgenic Mice: Effects on Organ and Skeletal Growth
Abstract
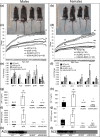
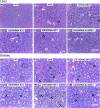
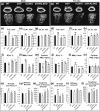
Growth hormone (GH) and insulinlike growth factor 1 (IGF-1) are anabolic hormones that facilitate somatic and skeletal growth and regulate metabolism via endocrine and autocrine/paracrine mechanisms. We hypothesized that excess tissue production of GH would protect skeletal growth and integrity in states of reduction in serum IGF-1 levels. To test our hypothesis, we used bovine GH (bGH) transgenic mice as a model of GH hypersecretion and ablated the liver-derived acid-labile subunit, which stabilizes IGF-1 complexes with IGF-binding protein-3 and -5 in circulation. We used a genetic approach to create bGH/als gene knockout (ALSKO) mice and small interfering RNA (siRNA) gene-silencing approach to reduce als or igf-1 gene expression. We found that in both models, decreased IGF-1 levels in serum were associated with decreased body and skeletal size of the bGH mice. Excess GH produced more robust bones but compromised mechanical properties in male mice. Excess GH production in tissues did not protect from trabecular bone loss in response to reductions in serum IGF-1 (in bGH/ALSKO or bGH mice treated with siRNAs). Reduced serum IGF-1 levels in the bGH mice did not alleviate the hyperinsulinemia and did not resolve liver or kidney pathologies that resulted from GH hypersecretion. We concluded that reduced serum IGF-1 levels decrease somatic and skeletal growth even in states of excess GH.
Copyright © 2017 Endocrine Society.
Figures



Similar articles
-
Lifelong Excess in GH Elicits Sexually Dimorphic Effects on Skeletal Morphology and Bone Mechanical Properties.J Bone Miner Res. 2022 Nov;37(11):2201-2214. doi: 10.1002/jbmr.4699. Epub 2022 Sep 16. J Bone Miner Res. 2022. PMID: 36069368 Free PMC article.
-
Skeletal response of male mice to anabolic hormone therapy in the absence of the Igfals gene.Endocrinology. 2014 Mar;155(3):987-99. doi: 10.1210/en.2013-1819. Epub 2014 Jan 1. Endocrinology. 2014. PMID: 24424061 Free PMC article.
-
Growth hormone mediates pubertal skeletal development independent of hepatic IGF-1 production.J Bone Miner Res. 2011 Apr;26(4):761-8. doi: 10.1002/jbmr.265. J Bone Miner Res. 2011. PMID: 20928887 Free PMC article.
-
A transgenic model to determine the physiological role of liver-derived insulin-like growth factor I.Minerva Endocrinol. 2002 Dec;27(4):299-311. Minerva Endocrinol. 2002. PMID: 12511852 Review.
-
A model for tissue-specific inducible insulin-like growth factor-I (IGF-I) inactivation to determine the physiological role of liver-derived IGF-I.Endocrine. 2002 Dec;19(3):249-56. doi: 10.1385/ENDO:19:3:249. Endocrine. 2002. PMID: 12624424 Review.
Cited by
-
Insulin-Like Growth Factors-1 Receptor (IGF-1R) Expression and the Phosphorylation of Endogenous Substrates Lead to Maturation of the Pacific oyster, Crassostrea gigas.Dev Reprod. 2021 Mar;25(1):67-72. doi: 10.12717/DR.2021.25.1.67. Epub 2021 Mar 31. Dev Reprod. 2021. PMID: 33977177 Free PMC article.
-
Mitochondrial Function Is Compromised in Cortical Bone Osteocytes of Long-Lived Growth Hormone Receptor Null Mice.J Bone Miner Res. 2019 Jan;34(1):106-122. doi: 10.1002/jbmr.3573. Epub 2018 Sep 14. J Bone Miner Res. 2019. PMID: 30216544 Free PMC article.
-
Multiplex PCR for the rapid detection of insulin-like growth factor in the Pacific oyster, Crassostrea gigas: a useful indicator for growth assessment.Mol Biol Rep. 2019 Feb;46(1):1023-1031. doi: 10.1007/s11033-018-4559-z. Epub 2018 Dec 13. Mol Biol Rep. 2019. PMID: 30547390
-
GH directly inhibits steatosis and liver injury in a sex-dependent and IGF1-independent manner.J Endocrinol. 2021 Jan;248(1):31-44. doi: 10.1530/JOE-20-0326. J Endocrinol. 2021. PMID: 33112796 Free PMC article.
-
Lifelong Excess in GH Elicits Sexually Dimorphic Effects on Skeletal Morphology and Bone Mechanical Properties.J Bone Miner Res. 2022 Nov;37(11):2201-2214. doi: 10.1002/jbmr.4699. Epub 2022 Sep 16. J Bone Miner Res. 2022. PMID: 36069368 Free PMC article.
References
-
- Feigerlova E, Hwa V, Derr MA, Rosenfeld RG. Current issues on molecular diagnosis of GH signaling defects. Endocr Dev. 2013;24:118–127. - PubMed
-
- Wit JM, Oostdijk W, Losekoot M. Spectrum of insulin-like growth factor deficiency. Endocr Dev. 2012;23:30–41. - PubMed
-
- Mohan S, Baylink DJ. IGF-binding proteins are multifunctional and act via IGF-dependent and -independent mechanisms. J Endocrinol. 2002;175(1):19–31. - PubMed
-
- Boisclair YR, Rhoads RP, Ueki I, Wang J, Ooi GT. The acid-labile subunit (ALS) of the 150 kDa IGF-binding protein complex: an important but forgotten component of the circulating IGF system. J Endocrinol. 2001;170(1):63–70. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

