Expression of progerin does not result in an increased mutation rate
- PMID: 28477268
- PMCID: PMC5662688
- DOI: 10.1007/s10577-017-9556-x
Expression of progerin does not result in an increased mutation rate
Abstract
In the premature ageing disease Hutchinson-Gilford progeria syndrome (HGPS), the underlying genetic defect in the lamin A gene leads to accumulation at the nuclear lamina of progerin-a mutant form of lamin A that cannot be correctly processed. This has been reported to result in defects in the DNA damage response and in DNA repair, leading to the hypothesis that, as in normal ageing and in other progeroid syndromes caused by mutation of genes of the DNA repair and DNA damage response pathways, increased DNA damage may be responsible for the premature ageing phenotypes in HGPS patients. However, this hypothesis is based upon the study of markers of the DNA damage response, rather than measurement of DNA damage per se or the consequences of unrepaired DNA damage-mutation. Here, using a mutation reporter cell line, we directly compared the inherent and induced mutation rates in cells expressing wild-type lamin A or progerin. We find no evidence for an elevated mutation rate in progerin-expressing cells. We conclude that the cellular defect in HGPS cells does not lie in the repair of DNA damage per se.
Keywords: DNA damage; Lamin A; nuclear lamina; progeria.
Figures
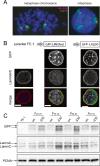
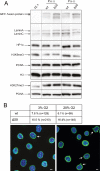
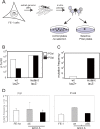

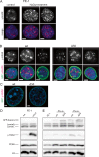
Similar articles
-
Epigenetic involvement in Hutchinson-Gilford progeria syndrome: a mini-review.Gerontology. 2014;60(3):197-203. doi: 10.1159/000357206. Epub 2014 Feb 28. Gerontology. 2014. PMID: 24603298 Review.
-
Identification of mitochondrial dysfunction in Hutchinson-Gilford progeria syndrome through use of stable isotope labeling with amino acids in cell culture.J Proteomics. 2013 Oct 8;91:466-77. doi: 10.1016/j.jprot.2013.08.008. Epub 2013 Aug 20. J Proteomics. 2013. PMID: 23969228
-
Heterochromatin loss as a determinant of progerin-induced DNA damage in Hutchinson-Gilford Progeria.Aging Cell. 2020 Mar;19(3):e13108. doi: 10.1111/acel.13108. Epub 2020 Feb 22. Aging Cell. 2020. PMID: 32087607 Free PMC article.
-
Progerin can induce DNA damage in the absence of global changes in replication or cell proliferation.PLoS One. 2024 Dec 5;19(12):e0315084. doi: 10.1371/journal.pone.0315084. eCollection 2024. PLoS One. 2024. PMID: 39636792 Free PMC article.
-
Towards delineating the chain of events that cause premature senescence in the accelerated aging syndrome Hutchinson-Gilford progeria (HGPS).Biochem Soc Trans. 2020 Jun 30;48(3):981-991. doi: 10.1042/BST20190882. Biochem Soc Trans. 2020. PMID: 32539085 Free PMC article. Review.
Cited by
-
Alteration of genetic recombination and double-strand break repair in human cells by progerin expression.DNA Repair (Amst). 2020 Dec;96:102975. doi: 10.1016/j.dnarep.2020.102975. Epub 2020 Sep 28. DNA Repair (Amst). 2020. PMID: 33010688 Free PMC article.
-
Nuclear lamina dysfunction triggers a germline stem cell checkpoint.Nat Commun. 2018 Sep 27;9(1):3960. doi: 10.1038/s41467-018-06277-z. Nat Commun. 2018. PMID: 30262885 Free PMC article.
-
Progerin impairs 3D genome organization and induces fragile telomeres by limiting the dNTP pools.Sci Rep. 2021 Jun 23;11(1):13195. doi: 10.1038/s41598-021-92631-z. Sci Rep. 2021. PMID: 34162976 Free PMC article.
References
-
- Bergo MO, Gavino B, Ross J, Schmidt WK, Hong C, Kendall LV, Mohr A, Meta M, Genant H, Jiang Y, et al. Zmpste24 deficiency in mice causes spontaneous bone fractures, muscle weakness, and a prelamin A processing defect. Proc Natl Acad Sci U S A. 2002;99:13049–13054. doi: 10.1073/pnas.192460799. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

