Thrombospondin-4 mediates TGF-β-induced angiogenesis
- PMID: 28481870
- PMCID: PMC5589494
- DOI: 10.1038/onc.2017.140
Thrombospondin-4 mediates TGF-β-induced angiogenesis
Abstract
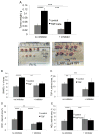
TGF-β is a multifunctional cytokine affecting many cell types and implicated in tissue remodeling processes. Due to its many functions and cell-specific effects, the consequences of TGF-β signaling are process-and stage-dependent, and it is not uncommon that TGF-β exerts distinct and sometimes opposing effects on a disease progression depending on the stage and on the pathological changes associated with the stage. The mechanisms underlying cell- and process-specific effects of TGF-β are poorly understood. We are describing a novel pathway that mediates induction of angiogenesis in response to TGF-β1. We found that in endothelial cells (EC) thrombospondin-4 (TSP-4), a secreted extracellular matrix (ECM) protein, is upregulated in response to TGF-β1 and mediates the effects of TGF-β1 on angiogenesis. Upregulation of TSP-4 does not require the synthesis of new protein, is not caused by decreased secretion of TSP-4, and is mediated by activation of SMAD3. Using Thbs4-/- mice and TSP-4 shRNA, we found that TSP-4 mediated pro-angiogenic functions in cultured EC and angiogenesis in vivo in response to TGF-β1. We observed~3-fold increases in tumor mass and levels of angiogenesis markers in animals injected with TGF-β1, and these effects did not occur in Thbs4-/- animals. Injections of an inhibitor of TGF-β1 signaling SB-431542 also decreased the weights of tumors and cancer angiogenesis. Our results from in vivo angiogenesis models and cultured EC document that TSP-4 mediates upregulation of angiogenesis by TGF-β1. Upregulation of pro-angiogenic TSP-4 and selective effects of TSP-4 on EC may contribute to stimulation of tumor growth by TGF-β despite the inhibition of cancer cell proliferation.
Conflict of interest statement
Figures




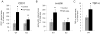


Similar articles
-
Autocrine thrombospondin partially mediates TGF-beta1- induced proliferation of vascular smooth muscle cells.Am J Physiol Heart Circ Physiol. 2000 Nov;279(5):H2159-65. doi: 10.1152/ajpheart.2000.279.5.H2159. Am J Physiol Heart Circ Physiol. 2000. PMID: 11045949
-
Thrombospondin-4 critically controls transforming growth factor β1 induced hypertrophic scar formation.J Cell Physiol. 2018 Jan;234(1):731-739. doi: 10.1002/jcp.26877. Epub 2018 Aug 21. J Cell Physiol. 2018. PMID: 30132849
-
Proangiogenic Properties of Thrombospondin-4.Arterioscler Thromb Vasc Biol. 2015 Sep;35(9):1975-86. doi: 10.1161/ATVBAHA.115.305912. Epub 2015 Jul 2. Arterioscler Thromb Vasc Biol. 2015. PMID: 26139464 Free PMC article.
-
Thrombospondin in renal disease.Nephron Exp Nephrol. 2009;111(3):e61-6. doi: 10.1159/000198235. Epub 2009 Jan 31. Nephron Exp Nephrol. 2009. PMID: 19182492 Review.
-
Regulation of tumor angiogenesis by thrombospondin-1.Biochim Biophys Acta. 2006 Apr;1765(2):178-88. doi: 10.1016/j.bbcan.2005.11.002. Epub 2005 Dec 21. Biochim Biophys Acta. 2006. PMID: 16406676 Review.
Cited by
-
The Activity of KIF14, Mieap, and EZR in a New Type of the Invasive Component, Torpedo-Like Structures, Predetermines the Metastatic Potential of Breast Cancer.Cancers (Basel). 2020 Jul 15;12(7):1909. doi: 10.3390/cancers12071909. Cancers (Basel). 2020. PMID: 32679794 Free PMC article.
-
TGF-β secreted by tumor-associated macrophages promotes proliferation and invasion of colorectal cancer via miR-34a-VEGF axis.Cell Cycle. 2018;17(24):2766-2778. doi: 10.1080/15384101.2018.1556064. Epub 2018 Dec 20. Cell Cycle. 2018. Retraction in: Cell Cycle. 2023 Mar-Mar;22(6):732. doi: 10.1080/15384101.2023.2171191. PMID: 30523755 Free PMC article. Retracted.
-
Transcriptome sequencing revealed that lymph node metastasis of papillary thyroid microcarcinoma is associated with high THBS4 expression and PDGFRA+ cancer-associated fibroblasts.Front Oncol. 2025 Apr 15;15:1536063. doi: 10.3389/fonc.2025.1536063. eCollection 2025. Front Oncol. 2025. PMID: 40303998 Free PMC article.
-
Pathological Significance and Prognostic Roles of Thrombospondin-3, 4 and 5 in Bladder Cancer.In Vivo. 2021 May-Jun;35(3):1693-1701. doi: 10.21873/invivo.12429. In Vivo. 2021. PMID: 33910854 Free PMC article.
-
Identification of candidate genes in ischemic cardiomyopathy by gene expression omnibus database.BMC Cardiovasc Disord. 2020 Jul 6;20(1):320. doi: 10.1186/s12872-020-01596-w. BMC Cardiovasc Disord. 2020. PMID: 32631246 Free PMC article.
References
-
- Lan HY, Chung AC. TGF-beta/Smad signaling in kidney disease. Semin Nephrol. 2012;32(3):236–43. - PubMed
-
- Fernandez IE, Eickelberg O. The impact of TGF-beta on lung fibrosis: from targeting to biomarkers. Proc Am Thorac Soc. 2012;9(3):111–6. - PubMed
-
- Weiss A, Attisano L. The TGFbeta superfamily signaling pathway. Wiley Interdiscip Rev Dev Biol. 2013;2(1):47–63. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

