Tight controlled dose reduction of biologics in psoriasis patients with low disease activity: a randomized pragmatic non-inferiority trial
- PMID: 28482858
- PMCID: PMC5422943
- DOI: 10.1186/s12895-017-0057-6
Tight controlled dose reduction of biologics in psoriasis patients with low disease activity: a randomized pragmatic non-inferiority trial
Abstract
Background: Psoriasis is an immune-mediated chronic inflammatory skin disorder for which several targeted biologic therapies became available in the last 10 years. Data from patients with rheumatoid arthritis revealed that dose tapering combined with tight control of disease activity is successful. For psoriasis patients the lowest effective dose of biologics needs to be determined. The objective was to assess whether dose tapering of biologics guided by Psoriasis Area and Severity Index (PASI) and Dermatology Quality of Life Index (DLQI) scores in psoriasis patients with controlled disease activity is non-inferior (NI) to usual care.
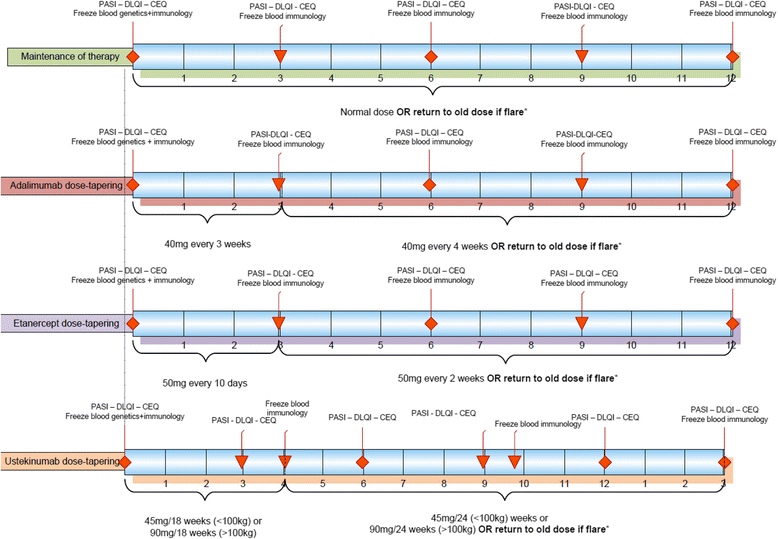
Methods/design: This is a multicenter, pragmatic, randomized, non-inferiority trial with cost- effectiveness analysis. One hundred and twenty patients with stable low disease activity (PASI ≤ 5 and DLQI ≤ 5) for at least 6 months with a stable use of adalimumab, etanercept or ustekinumab will be randomized 1:1 to the dose reduction group or usual care. In the dose reduction group, the treatment intervals will be prolonged stepwise, resulting in a 33% and 50% dose reduction, respectively. Disease activity is monitored every three months with PASI and DLQI. In case of flare the treatment is adjusted to the previous effective dose. The primary outcome (PASI) at 12 months will be analyzed with ANCOVA in which the baseline PASI will be included as covariate to gain efficiency. The secondary outcomes include number of and time to disease flares, health-related quality of life, serious adverse events, and costs.
Discussion: With this study we want to assess whether disease activity guided dose reduction of biologics can be achieved for psoriasis patients with low stable disease activity, without losing disease control. By using the lowest effective dose of biologics, we expect to minimize side effects and save costs.
Trial registration: This trial was registered at ClinicalTrials.gov ( NCT 02602925 ). Trial registration date October 9 2015.
Trial registration: ClinicalTrials.gov NCT02602925.
Keywords: Adalimumab; Biologics; Cost-effectiveness; Dose reduction; Etanercept; Non- inferiority; Psoriasis; Therapy; Ustekinumab.
Figures
Similar articles
-
Comparison of Tightly Controlled Dose Reduction of Biologics With Usual Care for Patients With Psoriasis: A Randomized Clinical Trial.JAMA Dermatol. 2020 Apr 1;156(4):393-400. doi: 10.1001/jamadermatol.2019.4897. JAMA Dermatol. 2020. PMID: 32049319 Free PMC article. Clinical Trial.
-
Dose reduction of the new generation biologics (IL-17 and IL-23 inhibitors) in psoriasis: study protocol for an international, pragmatic, multicenter, randomized, controlled, non-inferiority study-the BeNeBio study.Trials. 2021 Oct 16;22(1):707. doi: 10.1186/s13063-021-05681-z. Trials. 2021. PMID: 34656148 Free PMC article.
-
Two-year follow-up of a dose reduction strategy trial of biologics adalimumab, etanercept, and ustekinumab in psoriasis patients in daily practice.J Dermatolog Treat. 2022 May;33(3):1591-1597. doi: 10.1080/09546634.2020.1869147. Epub 2021 Jan 7. J Dermatolog Treat. 2022. PMID: 33356686 Clinical Trial.
-
Comparison of cumulative clinical benefits of biologics for the treatment of psoriasis over 16 weeks: Results from a network meta-analysis.J Am Acad Dermatol. 2020 May;82(5):1138-1149. doi: 10.1016/j.jaad.2019.12.038. Epub 2019 Dec 26. J Am Acad Dermatol. 2020. PMID: 31884091
-
Cost effectiveness of biologic therapies for plaque psoriasis.Am J Clin Dermatol. 2013 Aug;14(4):315-26. doi: 10.1007/s40257-013-0030-z. Am J Clin Dermatol. 2013. PMID: 23696234 Review.
Cited by
-
Comparison of Tightly Controlled Dose Reduction of Biologics With Usual Care for Patients With Psoriasis: A Randomized Clinical Trial.JAMA Dermatol. 2020 Apr 1;156(4):393-400. doi: 10.1001/jamadermatol.2019.4897. JAMA Dermatol. 2020. PMID: 32049319 Free PMC article. Clinical Trial.
-
Steps towards implementation of protocolized dose reduction of adalimumab, etanercept and ustekinumab for psoriasis in daily practice.J Dermatolog Treat. 2023 Dec;34(1):2186728. doi: 10.1080/09546634.2023.2186728. J Dermatolog Treat. 2023. PMID: 36867069 Free PMC article.
-
Pharmacogenetics Update on Biologic Therapy in Psoriasis.Medicina (Kaunas). 2020 Dec 20;56(12):719. doi: 10.3390/medicina56120719. Medicina (Kaunas). 2020. PMID: 33419370 Free PMC article. Review.
-
Lengthening adalimumab dosing interval in quiescent Crohn's disease patients: protocol for the pragmatic randomised non-inferiority LADI study.BMJ Open. 2020 May 26;10(5):e035326. doi: 10.1136/bmjopen-2019-035326. BMJ Open. 2020. PMID: 32461297 Free PMC article.
-
Health Economic Consequences of a Tightly Controlled Dose Reduction Strategy for Adalimumab, Etanercept and Ustekinumab Compared with Standard Psoriasis Care: A Cost-utility Analysis of the CONDOR Study.Acta Derm Venereol. 2020 Dec 1;100(19):adv00340. doi: 10.2340/00015555-3692. Acta Derm Venereol. 2020. PMID: 33196101 Free PMC article.
References
-
- Leonardi CL, Kimball AB, Papp KA, Yeilding N, Guzzo C, Wang Y, Li S, Dooley LT, Gordon KB. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 76-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 1) Lancet. 2008;371(9625):1665–1674. doi: 10.1016/S0140-6736(08)60725-4. - DOI - PubMed
-
- Gordon KB, Gottlieb AB, Langely RG, van de Kerkhof P, Belasco KT, Sundaram M, Okun M, Serra L. Adalimumab retreatment successfully restores clinical response and health-related quality of life in patients with moderate to severe psoriasis who undergo therapy interruption. J Eur Acad Dermatol Venereol. 2015;29(4):767–776. doi: 10.1111/jdv.12677. - DOI - PubMed
-
- Welsing PM, Bijl M, van Bodegraven AA, Lems WF, Prens E, Bijlsma JW. Cost effectiveness of biologicals: high costs are the other face of success. Ned Tijdschr Geneeskd. 2011;155(29):A3026. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

