Higher levels of myelin phospholipids in brains of neuronal α-Synuclein transgenic mice precede myelin loss
- PMID: 28482862
- PMCID: PMC5421332
- DOI: 10.1186/s40478-017-0439-3
Higher levels of myelin phospholipids in brains of neuronal α-Synuclein transgenic mice precede myelin loss
Abstract
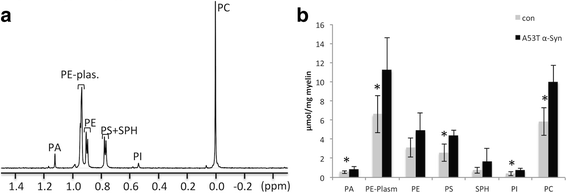
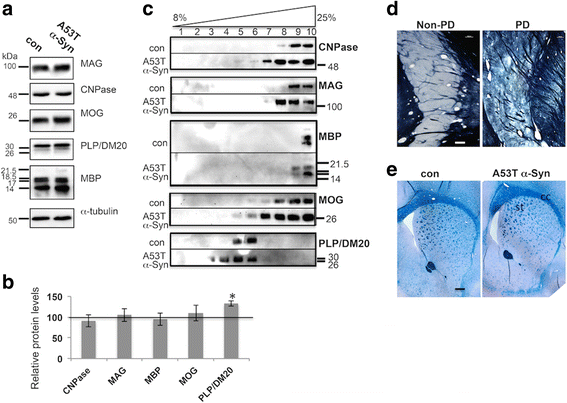
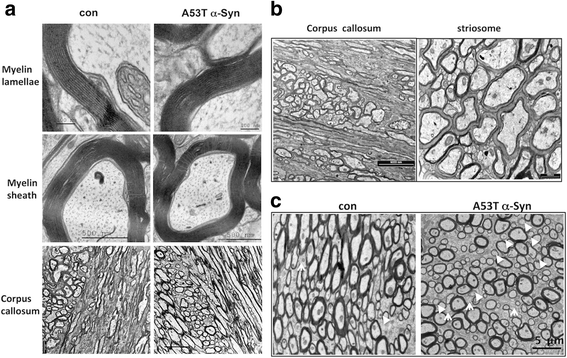
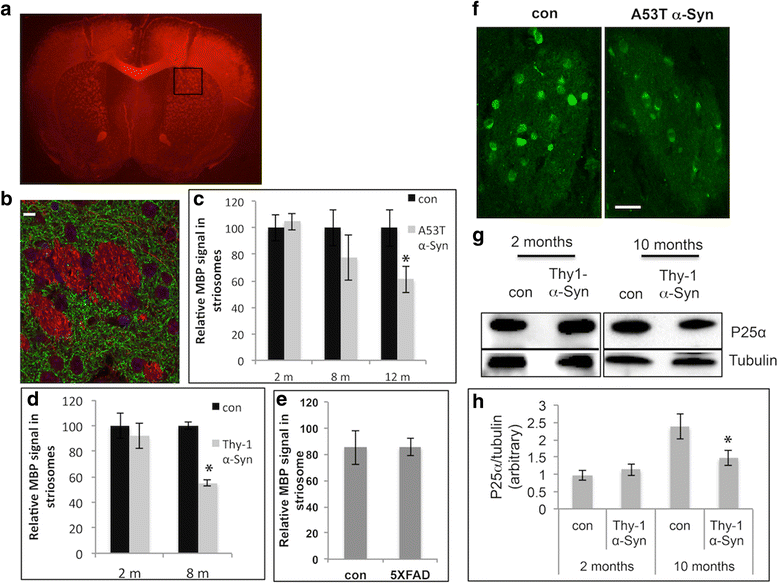
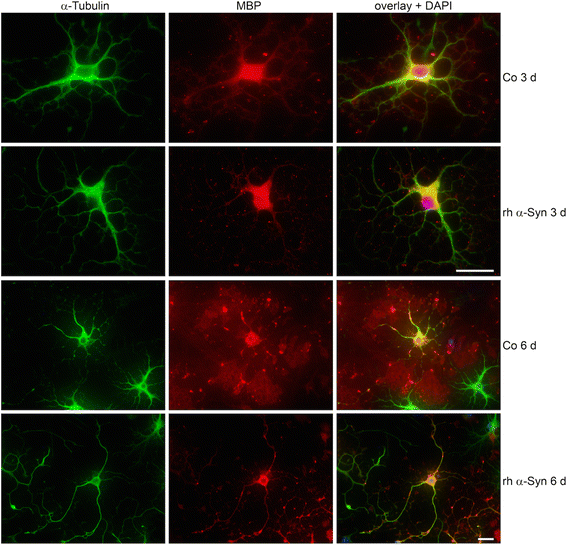
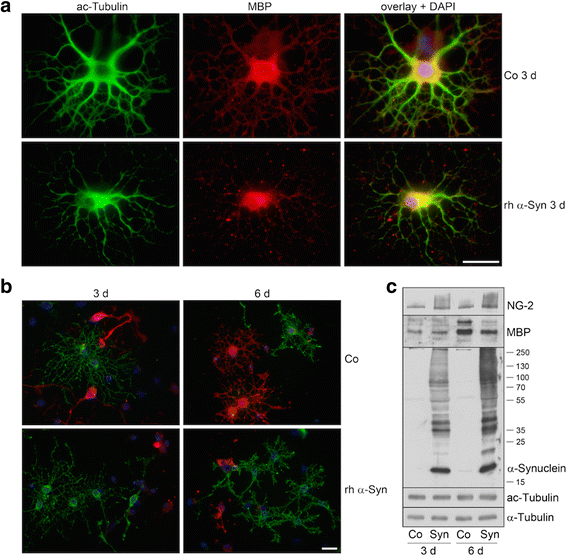
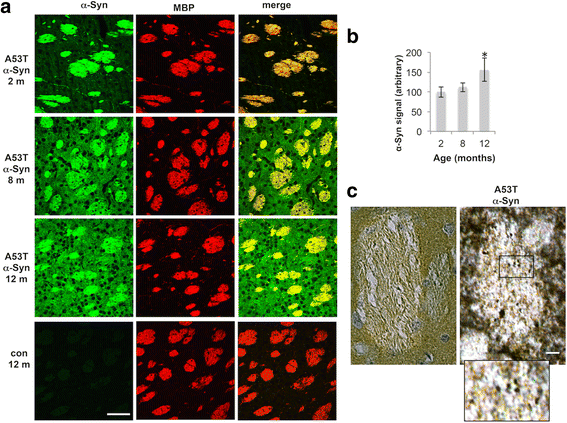
α-Synuclein is a protein involved in the pathogenesis of synucleinopathies, including Parkinson's disease (PD), dementia with Lewy bodies (DLB) and multiple system atrophy (MSA). We investigated the role of neuronal α-Syn in myelin composition and abnormalities. The phospholipid content of purified myelin was determined by 31P NMR in two mouse lines modeling PD, PrP-A53T α-Syn and Thy-1 wt-α-Syn. Significantly higher levels of phospholipids were detected in myelin purified from brains of these α-Syn transgenic mouse models than in control mice. Nevertheless, myelin ultrastructure appeared intact. To further investigate the effect of α-Syn on myelin abnormalities, we systematically analyzed the striatum, a brain region associated with neurodegeneration in PD. An age and disease-dependent loss of myelin basic protein (MBP) signal was detected by immunohistochemistry in striatal striosomes (patches). The age-dependent loss of MBP signal was associated with lower P25α levels in oligodendrocytes. In addition, we found that α-Syn inhibited oligodendrocyte maturation and the formation of membranous sheets in vitro. Based on these results we concluded that neuronal α-Syn is involved in the regulation and/or maintenance of myelin phospholipid. However, axonal hypomyelination in the PD models is evident only in progressive stages of the disease and associated with α-Syn toxicity.
Keywords: Myelin; Parkinson’s disease; Phospholipids; α-Synuclein.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

