The salvage therapy in lung adenocarcinoma initially harbored susceptible EGFR mutation and acquired resistance occurred to the first-line gefitinib and second-line cytotoxic chemotherapy
- PMID: 28486985
- PMCID: PMC5424380
- DOI: 10.1186/s40360-017-0130-0
The salvage therapy in lung adenocarcinoma initially harbored susceptible EGFR mutation and acquired resistance occurred to the first-line gefitinib and second-line cytotoxic chemotherapy
Abstract
Background: Epidermal growth factor receptor-tyrosine kinase inhibitors (EGFR-TKIs) such as gefitinib can provide better efficacy and prolonged progression free survival (PFS) than cytotoxic chemotherapy for metastatic lung non-squamous cell carcinoma harboring susceptible EGFR mutations when used as first-line therapy. Cytotoxic chemotherapy is regarded as being the standard therapy to overcome acquired resistance to an initial EGFR TKI. However, there is currently no consensus on how best to treat patients who develop resistance to both an initial EGFR TKI and chemotherapy.
Methods: We enrolled stage IV lung adenocarcinoma patients with an EGFR mutation and who had developed acquired resistance to gefitinib and cytotoxic chemotherapy from two university-affiliated hospitals in Taiwan from June 2011 to December 2014. Basic demographic data, included Eastern Cooperative Oncology Group (ECOG) performance status were collected, and the response rate, progression-free survival (PFS) and overall survival (OS) were analyzed.
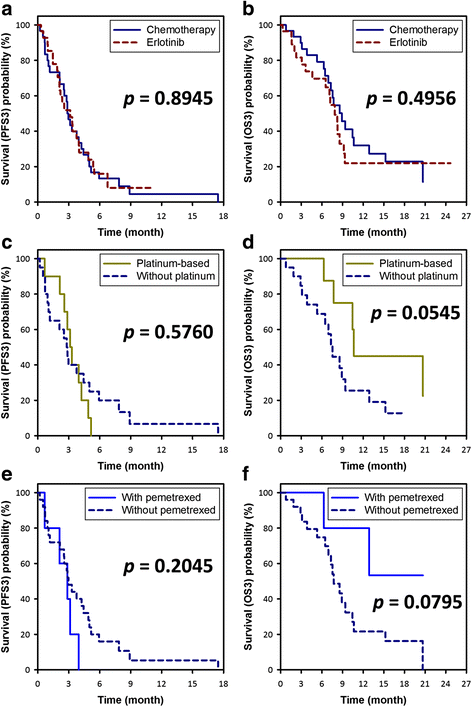
Result: Two hundred and nine patients with mutated EGFR and who took gefitinib as the first-line therapy were identified in the study period, of whom 86 received second-line cytotoxic chemotherapy, and 60 who received third-line therapy were eligible for this study. The patients who received cytotoxic chemotherapy had a significantly higher disease control rate than those who received erlotinib (73% vs. 46%, p = 0.0363), however there were no significant differences in PFS (2.9 months vs. 3.1 months, p = 0.9049) and OS (8.9 months vs. 7.9 months, p = 0.4956). Platinum- or pemetrexed-based chemotherapy provided similar PFS and OS as others did. The only significant poor prognostic factors for OS were old age (≥65 years) (HR = 5.97 [2.65-13.44], p < 0.0001) and poor performance status (ECOG ≥2) (HR = 5.84 [2.61-13.09], p < 0.0001).
Conclusion: Retreatment with an EGFR TKI is not inferior to cytotoxic chemotherapy when used as salvage therapy for patients with adenocarcinoma with an EGFR mutation, especially if a third-generation EGFR TKI is not available, or if the reason for resistance is unknown or is not related to the T790M mutation. Old age and poor ECOG score were both poor prognostic factors in the salvage therapy.
Keywords: Acquired resistance; Chemotherapy; Epidermal growth factor receptor; Erlotinib; Gefitinib.
Figures
References
-
- Paez JG, Janne PA, Lee JC, Tracy S, Greulich H, Gabriel S, Herman P, Kaye FJ, Lindeman N, Boggon TJ, Naoki K, Sasaki H, Fujii Y, Eck MJ, Sellers WR, Johnson BE, Meyerson M. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004;304:1497–1500. doi: 10.1126/science.1099314. - DOI - PubMed
-
- Rosell R, Carcereny E, Gervais R, Vergnenegre A, Massuti B, Felip E, Palmero R, Garcia-Gomez R, Pallares C, Sanchez JM, Porta R, Cobo M, Garrido P, Longo F, Moran T, Insa A, de Marinis F, Corre R, Bover I, Illiano A, Dansin E, De Castro J, Milella M, Reguart N, Altavilla G, Jimenez U, Provencio M, Moreno MA, Terrasa J, Munoz-Langa J, Valdivia J, Isla D, Domine M, Molinier O, Mazieres J, Baize N, Garcia-Campelo R, Robinet G, Rodriguez-Abreu D, Lopez-Vivanco G, Gebbia V, Ferrera-Delgado L, Bombaron P, Bernabe R, Bearz A, Artal A, Cortesi E, Rolfo C, Sanchez-Ronco M, Drozdowskyj A, Queralt C, de Aguirre I, Ramirez JL, Sanchez JJ, Molina MA, Taron M, Paz-Ares L. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012;13:239–246. doi: 10.1016/S1470-2045(11)70393-X. - DOI - PubMed
-
- Mitsudomi T, Morita S, Yatabe Y, Negoro S, Okamoto I, Tsurutani J, Seto T, Satouchi M, Tada H, Hirashima T, Asami K, Katakami N, Takada M, Yoshioka H, Shibata K, Kudoh S, Shimizu E, Saito H, Toyooka S, Nakagawa K, Fukuoka M. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomised phase 3 trial. Lancet Oncol. 2010;11:121–128. doi: 10.1016/S1470-2045(09)70364-X. - DOI - PubMed
-
- Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, Nishiwaki Y, Ohe Y, Yang JJ, Chewaskulyong B, Jiang H, Duffield EL, Watkins CL, Armour AA, Fukuoka M. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361:947–957. doi: 10.1056/NEJMoa0810699. - DOI - PubMed
-
- Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I, Fujita Y, Okinaga S, Hirano H, Yoshimori K, Harada T, Ogura T, Ando M, Miyazawa H, Tanaka T, Saijo Y, Hagiwara K, Morita S, Nukiwa T. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med. 2010;362:2380–2388. doi: 10.1056/NEJMoa0909530. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

