Apelin: A putative novel predictive biomarker for bevacizumab response in colorectal cancer
- PMID: 28487489
- PMCID: PMC5522118
- DOI: 10.18632/oncotarget.17306
Apelin: A putative novel predictive biomarker for bevacizumab response in colorectal cancer
Abstract
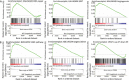
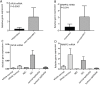
Bevacizumab (bvz) is currently employed as an anti-angiogenic therapy across several cancer indications. Bvz response heterogeneity has been well documented, with only 10-15% of colorectal cancer (CRC) patients benefitting in general. For other patients, clinical efficacy is limited and side effects are significant. This reinforces the need for a robust predictive biomarker of response. To identify such a biomarker, we performed a DNA microarray-based transcriptional profiling screen with primary endothelial cells (ECs) isolated from normal and tumour colon tissues. Thirteen separate populations of tumour-associated ECs and 10 of normal ECs were isolated using fluorescence-activated cell sorting. We hypothesised that VEGF-induced genes were overexpressed in tumour ECs; these genes could relate to bvz response and serve as potential predictive biomarkers. Transcriptional profiling revealed a total of 2,610 differentially expressed genes when tumour and normal ECs were compared. To explore their relation to bvz response, the mRNA expression levels of top-ranked genes were examined using quantitative PCR in 30 independent tumour tissues from CRC patients that received bvz in the adjuvant setting. These analyses revealed that the expression of MMP12 and APLN mRNA was significantly higher in bvz non-responders compared to responders. At the protein level, high APLN expression was correlated with poor progression-free survival in bvz-treated patients. Thus, high APLN expression may represent a novel predictive biomarker for bvz unresponsiveness.
Keywords: VEGF; apelin; bevacizumab response; biomarker; colorectal cancer.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Bergers G, Benjamin LE. Tumorigenesis and the angiogenic switch. Nat Rev Cancer. 2003;3:401–410. - PubMed
-
- Jubb AM, Hurwitz HI, Bai W, Holmgren EB, Tobin P, Guerrero AS, Kabbinavar F, Holden SN, Novotny WF, Frantz GD, Hillan KJ, Koeppen H. Impact of vascular endothelial growth factor-A expression, thrombospondin-2 expression, and microvessel density on the treatment effect of bevacizumab in metastatic colorectal cancer. J Clin Oncol. 2006;24:217–227. - PubMed
-
- Olsson AK, Dimberg A, Kreuger J, Claesson-Welsh L. VEGF receptor signalling - in control of vascular function. Nat Rev Mol Cell Biol. 2006;7:359–371. - PubMed
-
- Brufsky AM, Hurvitz S, Perez E, Swamy R, Valero V, O’Neill V, Rugo HS. RIBBON-2: a randomized, double-blind, placebo-controlled, phase III trial evaluating the efficacy and safety of bevacizumab in combination with chemotherapy for second-line treatment of human epidermal growth factor receptor 2-negative metastatic breast cancer. J Clin Oncol. 2011;29:4286–4293. - PubMed
-
- Friedman HS, Prados MD, Wen PY, Mikkelsen T, Schiff D, Abrey LE, Yung WK, Paleologos N, Nicholas MK, Jensen R, Vredenburgh J, Huang J, Zheng M, et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol. 2009;27:4733–4740. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

