Naoling decoction restores cognitive function by inhibiting the neuroinflammatory network in a rat model of Alzheimer's disease
- PMID: 28487495
- PMCID: PMC5522095
- DOI: 10.18632/oncotarget.17337
Naoling decoction restores cognitive function by inhibiting the neuroinflammatory network in a rat model of Alzheimer's disease
Abstract
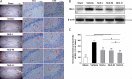
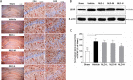
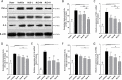
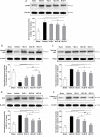
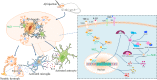
Neuroinflammation is central to the pathogenesis of Alzheimer's disease (AD). We previously showed that Naoling decoction (NLD), a traditional Chinese medicine, was effective against AD, acting by inhibiting expression of IL-1β and IL-6. In the present study, we generated the rat model of AD by injecting Aβ1-42 peptide intracerebroventricularly and evaluated the dose-dependent effects of NLD treatment. The NLD-treated rats exhibited significant improvements in cognitive function as evaluated by the Morris water maze test. Golgi-Cox staining revealed that NLD treatment dose-dependently increased dendritic spines in the CA1 region, which were diminished in vehicle-treated rats. Further, NLD treatment normalized hippocampal Chromogranin A levels, which were elevated by Aβ1-42 induction. NLD also attenuated activation of microglia and astrocytes induced by Aβ1-42. Subsequently, NLD dose-dependently reduced levels TNF-α, IL-1β and IL-6 by inhibiting the NF-κB signaling pathway and the ASC-dependent inflammasome in the hippocampus. These findings reveal that NLD is a promising therapeutic agent that exerts inhibitory effects at multiple sites within the neuroinflammatory network induced in AD.
Keywords: Alzheimer’s disease; Chromogranin A; Naoling decoction; amyloid-beta (Aβ) deposits; neuroinflammatory network.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Ritchie CW, Molinuevo JL, Truyen L, Satlin A, Van der Geyten S, Lovestone S. Development of interventions for the secondary prevention of Alzheimer's dementia: the European Prevention of Alzheimer's Dementia (EPAD) project. The Lancet Psychiatry. 2016;3:179–86. doi: 10.1016/s2215-0366(15)00454-x. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

