CaMKIIα may modulate fentanyl-induced hyperalgesia via a CeLC-PAG-RVM-spinal cord descending facilitative pain pathway in rats
- PMID: 28489932
- PMCID: PMC5425219
- DOI: 10.1371/journal.pone.0177412
CaMKIIα may modulate fentanyl-induced hyperalgesia via a CeLC-PAG-RVM-spinal cord descending facilitative pain pathway in rats
Abstract
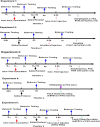
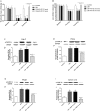
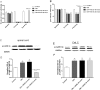
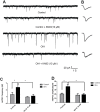
Each of the lateral capsular division of central nucleus of amygdala(CeLC), periaqueductal gray (PAG), rostral ventromedial medulla(RVM) and spinal cord has been proved to contribute to the development of opioid-induced hyperalgesia(OIH). Especially, Ca2+/calmodulin-dependent protein kinase IIα (CaMKIIα) in CeLC and spinal cord seems to play a key role in OIH modulation. However, the pain pathway through which CaMKIIα modulates OIH is not clear. The pathway from CeLC to spinal cord for this modulation was explored in the present study. Mechanical and thermal hyperalgesia were tested by von Frey test or Hargreaves test, respectively. CaMKIIα activity (phospho-CaMKIIα, p-CaMKIIα) was evaluated by western blot analysis. CaMKIIα antagonist (KN93) was micro-infused into CeLC, spinal cord or PAG, respectively, to evaluate its effect on behavioral hyperalgesia and p-CaMKIIα expression in CeLC, PAG, RVM and spinal cord. Then the underlying synaptic mechanism was explored by recording miniature excitatory postsynaptic currents (mEPSCs) on PAG slices using whole-cell voltage-clamp methods. Results showed that inhibition of CeLC, PAG or spinal CaMKIIα activity respectively by KN93, reversed both mechanical and thermal hyperalgesia. Microinjection of KN93 into CeLC decreased p-CaMKIIα expression in CeLC, PAG, RVM and spinal cord; while intrathecal KN93 can only block spinal but not CeLC CaMKIIα activity. KN93 injected into PAG just decreased p-CaMKIIα expression in PAG, RVM and spinal cord, but not in the CeLC. Similarly, whole-cell voltage-clamp recording found the frequency and amplitude of mEPSCs in PAG cells were decreased by KN93 added in PAG slice or micro-infused into CeLC in vivo. These results together with previous findings suggest that CaMKIIα may modulate OIH via a CeLC-PAG-RVM-spinal cord descending facilitative pain pathway.
Conflict of interest statement
Figures

Similar articles
-
Inhibition of CaMKIIα in the Central Nucleus of Amygdala Attenuates Fentanyl-Induced Hyperalgesia in Rats.J Pharmacol Exp Ther. 2016 Oct;359(1):82-9. doi: 10.1124/jpet.116.233817. Epub 2016 Jul 22. J Pharmacol Exp Ther. 2016. PMID: 27451410
-
[Role of mGluR5 in laterocapcular division of central nucleus of amygdala in fentanyl-induced hyperalgesia in rats].Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2019 Apr 28;44(4):364-369. doi: 10.11817/j.issn.1672-7347.2019.04.004. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2019. PMID: 31113910 Chinese.
-
Activation of the Extracellular Signal-Regulated Kinase in the Amygdale Modulates Fentanyl-Induced Hypersensitivity in Rats.J Pain. 2017 Feb;18(2):188-199. doi: 10.1016/j.jpain.2016.10.013. Epub 2016 Nov 10. J Pain. 2017. PMID: 27838497
-
The role of the periaqueductal gray in the modulation of pain in males and females: are the anatomy and physiology really that different?Neural Plast. 2009;2009:462879. doi: 10.1155/2009/462879. Epub 2009 Jan 28. Neural Plast. 2009. PMID: 19197373 Free PMC article. Review.
-
Endogenous opioid peptides in the descending pain modulatory circuit.Neuropharmacology. 2020 Aug 15;173:108131. doi: 10.1016/j.neuropharm.2020.108131. Epub 2020 May 15. Neuropharmacology. 2020. PMID: 32422213 Free PMC article. Review.
Cited by
-
HCN-Channel-Dependent Hyperexcitability of the Layer V Pyramidal Neurons in IL-mPFC Contributes to Fentanyl-Induced Hyperalgesia in Male Rats.Mol Neurobiol. 2023 May;60(5):2553-2571. doi: 10.1007/s12035-023-03218-w. Epub 2023 Jan 23. Mol Neurobiol. 2023. PMID: 36689134
-
Projections from infralimbic medial prefrontal cortex glutamatergic outputs to amygdala mediates opioid induced hyperalgesia in male rats.Mol Pain. 2024 Jan-Dec;20:17448069241226960. doi: 10.1177/17448069241226960. Mol Pain. 2024. PMID: 38172075 Free PMC article.
-
The Dynorphin/-Opioid Receptor System at the Interface of Hyperalgesia/Hyperkatifeia and Addiction.Curr Addict Rep. 2025;12(1):11. doi: 10.1007/s40429-025-00618-x. Epub 2025 Feb 4. Curr Addict Rep. 2025. PMID: 40124896 Free PMC article. Review.
-
The rising crisis of illicit fentanyl use, overdose, and potential therapeutic strategies.Transl Psychiatry. 2019 Nov 11;9(1):282. doi: 10.1038/s41398-019-0625-0. Transl Psychiatry. 2019. PMID: 31712552 Free PMC article. Review.
-
Restoration of the Activity of the Prefrontal Cortex to the Nucleus Accumbens Core Pathway Relieves Fentanyl-Induced Hyperalgesia in Male Rats.J Pain Res. 2024 Mar 20;17:1243-1256. doi: 10.2147/JPR.S442765. eCollection 2024. J Pain Res. 2024. PMID: 38524691 Free PMC article.
References
-
- Mao J. Opioid-induced abnormal pain sensitivity: implications in clinical opioid therapy. Pain. 2002;100(3):213–7. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

