Inflammatory cytokines down-regulate the barrier-protective prostasin-matriptase proteolytic cascade early in experimental colitis
- PMID: 28490634
- PMCID: PMC5491767
- DOI: 10.1074/jbc.M116.771469
Inflammatory cytokines down-regulate the barrier-protective prostasin-matriptase proteolytic cascade early in experimental colitis
Abstract
Compromised gastrointestinal barrier function is strongly associated with the progressive and destructive pathologies of the two main forms of irritable bowel disease (IBD), ulcerative colitis (UC), and Crohn's disease (CD). Matriptase is a membrane-anchored serine protease encoded by
Keywords: cell junction; colitis; cytokine; inflammatory bowel disease (IBD); intestinal epithelium; matriptase; permeability; prostasin; protease; serine protease.
© 2017 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures

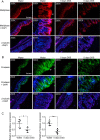
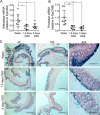
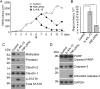
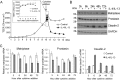
Similar articles
-
Matriptase protects against experimental colitis and promotes intestinal barrier recovery.Inflamm Bowel Dis. 2012 Jul;18(7):1303-14. doi: 10.1002/ibd.21930. Epub 2011 Nov 13. Inflamm Bowel Dis. 2012. PMID: 22081509 Free PMC article.
-
STAT6 activation in ulcerative colitis: a new target for prevention of IL-13-induced colon epithelial cell dysfunction.Inflamm Bowel Dis. 2011 Nov;17(11):2224-34. doi: 10.1002/ibd.21628. Epub 2011 Feb 9. Inflamm Bowel Dis. 2011. PMID: 21308881 Free PMC article.
-
Sodium absorption stimulator prostasin (PRSS8) has an anti-inflammatory effect via downregulation of TLR4 signaling in inflammatory bowel disease.J Gastroenterol. 2020 Apr;55(4):408-417. doi: 10.1007/s00535-019-01660-z. Epub 2020 Jan 8. J Gastroenterol. 2020. PMID: 31916038
-
The matriptase-prostasin proteolytic cascade in epithelial development and pathology.Cell Tissue Res. 2013 Feb;351(2):245-53. doi: 10.1007/s00441-012-1348-1. Epub 2012 Feb 18. Cell Tissue Res. 2013. PMID: 22350849 Review.
-
Beneficial effects of nutritional supplements on intestinal epithelial barrier functions in experimental colitis models in vivo.World J Gastroenterol. 2019 Aug 14;25(30):4181-4198. doi: 10.3748/wjg.v25.i30.4181. World J Gastroenterol. 2019. PMID: 31435172 Free PMC article. Review.
Cited by
-
Time-dependent ultrastructural changes during venous thrombogenesis and thrombus resolution.J Thromb Haemost. 2024 Jun;22(6):1675-1688. doi: 10.1016/j.jtha.2024.02.020. Epub 2024 Mar 15. J Thromb Haemost. 2024. PMID: 38492853 Free PMC article.
-
The Effect of Serine Protease Inhibitors on Visceral Pain in Different Rodent Models With an Intestinal Insult.Front Pharmacol. 2022 Jun 2;13:765744. doi: 10.3389/fphar.2022.765744. eCollection 2022. Front Pharmacol. 2022. PMID: 35721192 Free PMC article.
-
Signal transducer and activator of transcription 6 as a target in colon cancer therapy.Oncol Lett. 2020 Jul;20(1):455-464. doi: 10.3892/ol.2020.11614. Epub 2020 May 13. Oncol Lett. 2020. PMID: 32565970 Free PMC article. Review.
-
Acetylcholinesterase Inhibitor Pyridostigmine Bromide Attenuates Gut Pathology and Bacterial Dysbiosis in a Murine Model of Ulcerative Colitis.Dig Dis Sci. 2020 Jan;65(1):141-149. doi: 10.1007/s10620-019-05838-6. Epub 2019 Oct 23. Dig Dis Sci. 2020. PMID: 31643033 Free PMC article.
-
Downregulation of Matriptase Inhibits Porphyromonas gingivalis Lipopolysaccharide-Induced Matrix Metalloproteinase-1 and Proinflammatory Cytokines by Suppressing the TLR4/NF-κB Signaling Pathways in Human Gingival Fibroblasts.Biomed Res Int. 2022 Oct 4;2022:3865844. doi: 10.1155/2022/3865844. eCollection 2022. Biomed Res Int. 2022. PMID: 36246974 Free PMC article.
References
-
- Xavier R. J., and Podolsky D. K. (2007) Unravelling the pathogenesis of inflammatory bowel disease. Nature 448, 427–434 - PubMed
-
- Hollander D., Vadheim C. M., Brettholz E., Petersen G. M., Delahunty T., and Rotter J. I. (1986) Increased intestinal permeability in patients with Crohn's disease and their relatives. A possible etiologic factor. Ann. Intern. Med. 105, 883–885 - PubMed
-
- Katz K. D., Hollander D., Vadheim C. M., McElree C., Delahunty T., Dadufalza V. D., Krugliak P., and Rotter J. I. (1989) Intestinal permeability in patients with Crohn's disease and their healthy relatives. Gastroenterology 97, 927–931 - PubMed
-
- Irvine E. J., and Marshall J. K. (2000) Increased intestinal permeability precedes the onset of Crohn's disease in a subject with familial risk. Gastroenterology 119, 1740–1744 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

