Dendritic cells provide a therapeutic target for synthetic small molecule analogues of the parasitic worm product, ES-62
- PMID: 28490801
- PMCID: PMC5431997
- DOI: 10.1038/s41598-017-01651-1
Dendritic cells provide a therapeutic target for synthetic small molecule analogues of the parasitic worm product, ES-62
Abstract
ES-62, a glycoprotein secreted by the parasitic filarial nematode Acanthocheilonema viteae, subverts host immune responses towards anti-inflammatory phenotypes by virtue of covalently attached phosphorylcholine (PC). The PC dictates that ES-62 exhibits protection in murine models of inflammatory disease and hence a library of drug-like PC-based small molecule analogues (SMAs) was synthesised. Four sulfone-containing SMAs termed 11a, 11e, 11i and 12b were found to reduce mouse bone marrow-derived dendritic cell (DC) pathogen-associated molecular pattern (PAMP)-induced pro-inflammatory cytokine production, inhibit NF-κB p65 activation, and suppress LPS-induced up-regulation of CD40 and CD86. Active SMAs also resulted in a DC phenotype that exhibited reduced capacity to prime antigen (Ag)-specific IFN-γ production during co-culture with naïve transgenic TCR DO.11.10 T cells in vitro and reduced their ability, following adoptive transfer, to prime the expansion of Ag-specific T lymphocytes, specifically TH17 cells, in vivo. Consistent with this, mice receiving DCs treated with SMAs exhibited significantly reduced severity of collagen-induced arthritis and this was accompanied by a significant reduction in IL-17+ cells in the draining lymph nodes. Collectively, these studies indicate that drug-like compounds that target DCs can be designed from parasitic worm products and demonstrate the potential for ES-62 SMA-based DC therapy in inflammatory disease.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
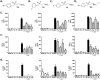
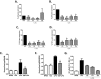

 p < 0.05 compared to OVA DCs and
p < 0.05 compared to OVA DCs and  p < 0.05 and
p < 0.05 and  p < 0.01 compared to OVA+LPS DCs.
p < 0.01 compared to OVA+LPS DCs.
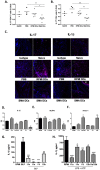
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

