Development and evaluation of a new modular nanotransporter for drug delivery into nuclei of pathological cells expressing folate receptors
- PMID: 28490863
- PMCID: PMC5413543
- DOI: 10.2147/DDDT.S127270
Development and evaluation of a new modular nanotransporter for drug delivery into nuclei of pathological cells expressing folate receptors
Abstract
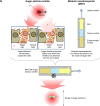
Purpose: Modular nanotransporters (MNTs) are artificial multifunctional systems designed to facilitate receptor-specific transport from the cell surface into the cell nucleus through inclusion of polypeptide domains for accomplishing receptor binding and internalization, as well as sequential endosomal escape and nuclear translocation. The objective of this study was to develop a new MNT targeted at folate receptors (FRs) for precise delivery of therapeutic cargo to the nuclei of FR-positive cells and to evaluate its potential, particularly for delivery of therapeutic agents (eg, the Auger electron emitter 111In) into the nuclei of target cancer cells.
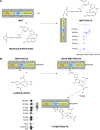
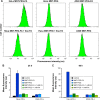
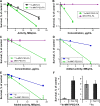
Methods: A FR-targeted MNT was developed by site-specific derivatization of ligand-free MNT with maleimide-polyethylene glycol-folic acid. The ability of FR-targeted MNT to accumulate in target FR-expressing cells was evaluated using flow cytometry, and intracellular localization of this MNT was assessed using confocal laser scanning microscopy of cells. The cytotoxicity of the 111In-labeled FR-targeted MNT was evaluated on HeLa and U87MG cancer cell lines expressing FR. In vivo micro-single-photon emission computed tomography/CT imaging and antitumor efficacy studies were performed with intratumoral injection of 111In-labeled FR-targeted MNT in HeLa xenograft-bearing mice.
Results: The resulting FR-targeted MNT accumulated in FR-positive HeLa cancer cell lines specifically and demonstrated the ability to reach its target destination - the cell nuclei. 111In-labeled FR-targeted MNT demonstrated efficient and specific FR-positive cancer cell eradication. A HeLa xenograft in vivo model revealed prolonged retention of 111In delivered by FR-targeted MNT and significant tumor growth delay (up to 80% growth inhibition).
Conclusion: The FR-targeted MNT met expectations of its ability to deliver active cargo into the nuclei of target FR-positive cells efficiently and specifically. As a result of this finding the new FR-targeted MNT approach warrants broad evaluation.
Keywords: cancer; folic acid; indium-111; nuclear delivery; radionuclide therapy.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures



Similar articles
-
Modular Nanotransporters for Nuclear-Targeted Delivery of Auger Electron Emitters.Front Pharmacol. 2018 Aug 27;9:952. doi: 10.3389/fphar.2018.00952. eCollection 2018. Front Pharmacol. 2018. PMID: 30210340 Free PMC article. Review.
-
Preparation, cytotoxicity, and in vivo antitumor efficacy of 111In-labeled modular nanotransporters.Int J Nanomedicine. 2017 Jan 10;12:395-410. doi: 10.2147/IJN.S125359. eCollection 2017. Int J Nanomedicine. 2017. PMID: 28138237 Free PMC article.
-
Antitumor efficacy of Auger electron emitter 111In delivered by modular nanotransporter into the nuclei of cells with folate receptor overexpression.Dokl Biochem Biophys. 2017 Mar;473(1):85-87. doi: 10.1134/S1607672917020016. Epub 2017 May 17. Dokl Biochem Biophys. 2017. PMID: 28510138
-
Modular nanotransporters: a multipurpose in vivo working platform for targeted drug delivery.Int J Nanomedicine. 2012;7:467-82. doi: 10.2147/IJN.S28249. Epub 2012 Feb 2. Int J Nanomedicine. 2012. PMID: 22346349 Free PMC article.
-
The folate receptor as a rational therapeutic target for personalized cancer treatment.Drug Resist Updat. 2014 Oct-Dec;17(4-6):89-95. doi: 10.1016/j.drup.2014.10.002. Epub 2014 Oct 8. Drug Resist Updat. 2014. PMID: 25457975 Review.
Cited by
-
Advancements in the use of Auger electrons in science and medicine during the period 2015-2019.Int J Radiat Biol. 2023;99(1):2-27. doi: 10.1080/09553002.2020.1831706. Epub 2020 Oct 23. Int J Radiat Biol. 2023. PMID: 33021416 Free PMC article. Review.
-
New Recombinant Carriers Binding Specifically to the Epidermal Growth Factor Receptor.Dokl Biochem Biophys. 2020 Jan;490(1):22-24. doi: 10.1134/S1607672920010081. Epub 2020 Apr 27. Dokl Biochem Biophys. 2020. PMID: 32342307
-
Prospects of Using Protein Engineering for Selective Drug Delivery into a Specific Compartment of Target Cells.Pharmaceutics. 2023 Mar 19;15(3):987. doi: 10.3390/pharmaceutics15030987. Pharmaceutics. 2023. PMID: 36986848 Free PMC article. Review.
-
Subcellular Targeting of Theranostic Radionuclides.Front Pharmacol. 2018 Sep 4;9:996. doi: 10.3389/fphar.2018.00996. eCollection 2018. Front Pharmacol. 2018. PMID: 30233374 Free PMC article. Review.
-
Modular Nanotransporters for Nuclear-Targeted Delivery of Auger Electron Emitters.Front Pharmacol. 2018 Aug 27;9:952. doi: 10.3389/fphar.2018.00952. eCollection 2018. Front Pharmacol. 2018. PMID: 30210340 Free PMC article. Review.
References
-
- Tyagarajan K, Pretzer E, Wiktorowicz JE. Thiol-reactive dyes for fluorescence labeling of proteomic samples. Electrophoresis. 2003;24(14):2348–2358. - PubMed
-
- Shafer DE, Inman JK, Lees A. Reaction of Tris(2-carboxyethyl)phosphine (TCEP) with maleimide and α-haloacyl groups: anomalous elution of TCEP by gel filtration. Anal Biochem. 2000;282(1):161–164. - PubMed
-
- Lu Y, Low PS. Folate-mediated delivery of macromolecular anticancer therapeutic agents. Adv Drug Deliv Rev. 2002;54(5):675–693. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

