Na +/Ca2+ exchangers and Orai channels jointly refill endoplasmic reticulum (ER) Ca2+ via ER nanojunctions in vascular endothelial cells
- PMID: 28497275
- PMCID: PMC5590033
- DOI: 10.1007/s00424-017-1989-8
Na +/Ca2+ exchangers and Orai channels jointly refill endoplasmic reticulum (ER) Ca2+ via ER nanojunctions in vascular endothelial cells
Abstract
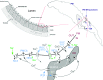
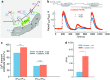
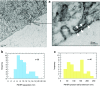
We investigated the role of Na+/ Ca2+ exchange (NCX) in the refilling of endoplasmic reticulum (ER) Ca2+ in vascular endothelial cells under various conditions of cell stimulation and plasma membrane (PM) polarization. Better understanding of the mechanisms behind basic ER Ca2+ content regulation is important, since current hypotheses on the possible ultimate causes of ER stress point to deterioration of the Ca2+ transport mechanism to/from ER itself. We measured [Ca2+]i temporal changes by Fura-2 fluorescence under experimental protocols that inhibit a host of transporters (NCX, Orai, non-selective transient receptor potential canonical (TRPC) channels, sarco/endoplasmic reticulum Ca2+ ATPase (SERCA), Na+/ K+ ATPase (NKA)) involved in the Ca2+ communication between the extracellular space and the ER. Following histamine-stimulated ER Ca2+ release, blockade of NCX Ca2+-influx mode (by 10 μM KB-R7943) diminished the ER refilling capacity by about 40%, while in Orai1 dominant negative-transfected cells NCX blockade attenuated ER refilling by about 60%. Conversely, inhibiting the ouabain sensitive NKA (10 nM ouabain), which may be localized in PM-ER junctions, increased the ER Ca2+ releasable fraction by about 20%, thereby supporting the hypothesis that this process of privileged ER refilling is junction-mediated. Junctions were observed in the cell ultrastructure and their main parameters of membrane separation and linear extension were (9.6 ± 3.8) nm and (128 ± 63) nm, respectively. Our findings point to a process of privileged refilling of the ER, in which NCX and store-operated Ca2+ entry via the stromal interaction molecule (STIM)-Orai system are the sole protagonists. These results shed light on the molecular machinery involved in the function of a previously hypothesized subplasmalemmal Ca2+ control unit during ER refilling with extracellular Ca2+.
Keywords: Calcium signals; EA.hy926; ER junctions; Endothelium; NCX; Orai1.
Figures




Similar articles
-
Privileged coupling between Ca(2+) entry through plasma membrane store-operated Ca(2+) channels and the endoplasmic reticulum Ca(2+) pump.Mol Cell Endocrinol. 2012 Apr 28;353(1-2):37-44. doi: 10.1016/j.mce.2011.08.021. Epub 2011 Aug 24. Mol Cell Endocrinol. 2012. PMID: 21878366 Review.
-
Three-dimensional spatio-temporal modelling of store operated Ca2+ entry: Insights into ER refilling and the spatial signature of Ca2+ signals.Cell Calcium. 2018 Jul;73:11-24. doi: 10.1016/j.ceca.2018.03.006. Epub 2018 Apr 4. Cell Calcium. 2018. PMID: 29880194
-
TRPC1, STIM1, and ORAI influence signal-regulated intracellular and endoplasmic reticulum calcium dynamics in human myometrial cells.Biol Reprod. 2011 Aug;85(2):315-26. doi: 10.1095/biolreprod.111.091082. Epub 2011 May 12. Biol Reprod. 2011. PMID: 21565997 Free PMC article.
-
Ca(2+) removal mechanisms in freshly isolated rabbit aortic endothelial cells.Cell Calcium. 2002 Jun;31(6):265-77. doi: 10.1016/s0143-4160(02)00075-1. Cell Calcium. 2002. PMID: 12098216
-
Regulation and Role of Store-Operated Ca2+ Entry in Cellular Proliferation.In: Kozak JA, Putney JW Jr, editors. Calcium Entry Channels in Non-Excitable Cells. Boca Raton (FL): CRC Press/Taylor & Francis; 2018. Chapter 12. In: Kozak JA, Putney JW Jr, editors. Calcium Entry Channels in Non-Excitable Cells. Boca Raton (FL): CRC Press/Taylor & Francis; 2018. Chapter 12. PMID: 30299656 Free Books & Documents. Review.
Cited by
-
Reactive Oxygen Species and Endothelial Ca2+ Signaling: Brothers in Arms or Partners in Crime?Int J Mol Sci. 2021 Sep 10;22(18):9821. doi: 10.3390/ijms22189821. Int J Mol Sci. 2021. PMID: 34575985 Free PMC article. Review.
-
The Molecular Heterogeneity of Store-Operated Ca2+ Entry in Vascular Endothelial Cells: The Different roles of Orai1 and TRPC1/TRPC4 Channels in the Transition from Ca2+-Selective to Non-Selective Cation Currents.Int J Mol Sci. 2023 Feb 7;24(4):3259. doi: 10.3390/ijms24043259. Int J Mol Sci. 2023. PMID: 36834672 Free PMC article. Review.
-
Modeling of Ca2+ transients initiated by GPCR agonists in mesenchymal stromal cells.BBA Adv. 2021 Apr 21;1:100012. doi: 10.1016/j.bbadva.2021.100012. eCollection 2021. BBA Adv. 2021. PMID: 37082025 Free PMC article.
-
Hydrogen Sulfide-Evoked Intracellular Ca2+ Signals in Primary Cultures of Metastatic Colorectal Cancer Cells.Cancers (Basel). 2020 Nov 11;12(11):3338. doi: 10.3390/cancers12113338. Cancers (Basel). 2020. PMID: 33187307 Free PMC article.
-
α-Latrotoxin Actions in the Absence of Extracellular Ca2+ Require Release of Stored Ca2.Toxins (Basel). 2025 Feb 6;17(2):73. doi: 10.3390/toxins17020073. Toxins (Basel). 2025. PMID: 39998090 Free PMC article.
References
-
- Andrikopoulos P, Baba A, Matsuda T, Djamgoz MBA, Yaqoob MM, Eccles SA. Ca2+ influx through reverse mode na+/ca2+ exchange is critical for vascular endothelial growth factor-mediated extracellular signal-regulated kinase (erk) 1/2 activation and angiogenic functions of human endothelial cells. J Biol Chem. 2011;286(44):37919–37931. doi: 10.1074/jbc.M111.251777. - DOI - PMC - PubMed
-
- Arnon A, Hamlyn JM, Blaustein MP. Ouabain augments ca(2+) transients in arterial smooth muscle without raising cytosolic na(+) Am J Physiol Heart Circ Physiol. 2000;279(2):H679–H691. - PubMed
-
- Blaustein MP, Zhang J, Chen L, Song H, Raina H, Kinsey SP, Izuka M, Iwamoto T, Kotlikoff MI, Lingrel JB, Philipson KD, Wier WG, Hamlyn JM. The pump, the exchanger, and endogenous ouabain: signaling mechanisms that link salt retention to hypertension. Hypertension. 2009;53(2):291–298. doi: 10.1161/HYPERTENSIONAHA.108.119974. - DOI - PMC - PubMed
-
- Bondarenko AI, Drachuk K, Panasiuk O, Sagach V, Deak AT, Malli R, Graier WF. N-arachidonoyl glycine suppresses na/ca2 exchanger-mediated ca2 entry into endothelial cells and activates bk(ca) channels independently of gpcrs. Br J Pharmacol. 2013;169(4):933–948. doi: 10.1111/bph.12180. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

