Microgravity induces inhibition of osteoblastic differentiation and mineralization through abrogating primary cilia
- PMID: 28500304
- PMCID: PMC5431935
- DOI: 10.1038/s41598-017-02049-9
Microgravity induces inhibition of osteoblastic differentiation and mineralization through abrogating primary cilia
Abstract
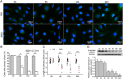
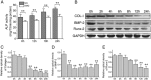
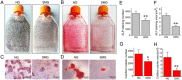
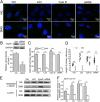
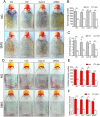
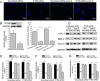
It is well documented that microgravity in space environment leads to bone loss in astronauts. These physiological changes have also been validated by human and animal studies and modeled in cell-based analogs. However, the underlying mechanisms are elusive. In the current study, we identified a novel phenomenon that primary cilia (key sensors and functioning organelles) of rat calvarial osteoblasts (ROBs) gradually shrank and disappeared almost completely after exposure to simulated microgravity generated by a random positioning machine (RPM). Along with the abrogation of primary cilia, the differentiation, maturation and mineralization of ROBs were inhibited. We also found that the disappearance of primary cilia was prevented by treating ROBs with cytochalasin D, but not with LiCl or dynein light chain Tctex-type 1 (Dynlt1) siRNA. The repression of the differentiation, maturation and mineralization of ROBs was effectively offset by cytochalasin D treatment in microgravity conditions. Blocking ciliogenesis using intraflagellar transport protein 88 (IFT88) siRNA knockdown inhibited the ability of cytochalasin D to counteract this reduction of osteogenesis. These results indicate that the abrogation of primary cilia may be responsible for the microgravity's inhibition on osteogenesis. Reconstruction of primary cilia may become a potential strategy against bone loss induced by microgravity.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
Protection of primary cilia is an effective countermeasure against the impairment of osteoblast function induced by simulated microgravity.J Cell Mol Med. 2023 Jan;27(1):36-51. doi: 10.1111/jcmm.17628. Epub 2022 Dec 13. J Cell Mol Med. 2023. PMID: 36512344 Free PMC article.
-
Primary cilia act as microgravity sensors by depolymerizing microtubules to inhibit osteoblastic differentiation and mineralization.Bone. 2020 Jul;136:115346. doi: 10.1016/j.bone.2020.115346. Epub 2020 Mar 30. Bone. 2020. PMID: 32240849
-
Simulated microgravity-induced oxidative stress and loss of osteogenic potential of osteoblasts can be prevented by protection of primary cilia.J Cell Physiol. 2023 Nov;238(11):2692-2709. doi: 10.1002/jcp.31127. Epub 2023 Oct 5. J Cell Physiol. 2023. PMID: 37796139
-
[Mechanisms of bone formation by primary cilia].Nihon Yakurigaku Zasshi. 2024 Jul 1;159(4):198-202. doi: 10.1254/fpj.23113. Epub 2024 Apr 26. Nihon Yakurigaku Zasshi. 2024. PMID: 38684398 Review. Japanese.
-
The effect of microgravity on morphology and gene expression of osteoblasts in vitro.FASEB J. 1999;13 Suppl:S129-34. doi: 10.1096/fasebj.13.9001.s129. FASEB J. 1999. PMID: 10352154 Review.
Cited by
-
Protection of primary cilia is an effective countermeasure against the impairment of osteoblast function induced by simulated microgravity.J Cell Mol Med. 2023 Jan;27(1):36-51. doi: 10.1111/jcmm.17628. Epub 2022 Dec 13. J Cell Mol Med. 2023. PMID: 36512344 Free PMC article.
-
Osteogenic Stimulation of Human Adipose-Derived Mesenchymal Stem Cells Using a Fungal Metabolite That Suppresses the Polycomb Group Protein EZH2.Stem Cells Transl Med. 2018 Feb;7(2):197-209. doi: 10.1002/sctm.17-0086. Epub 2017 Dec 27. Stem Cells Transl Med. 2018. PMID: 29280310 Free PMC article.
-
Melatonin Regulates Osteoblast Differentiation through the m6A Reader hnRNPA2B1 under Simulated Microgravity.Curr Issues Mol Biol. 2024 Sep 1;46(9):9624-9638. doi: 10.3390/cimb46090572. Curr Issues Mol Biol. 2024. PMID: 39329924 Free PMC article.
-
Immunity in Space: Prokaryote Adaptations and Immune Response in Microgravity.Life (Basel). 2021 Feb 2;11(2):112. doi: 10.3390/life11020112. Life (Basel). 2021. PMID: 33540536 Free PMC article. Review.
-
Large-scale analysis reveals spatiotemporal circadian patterns of cilia transcriptomes in the primate brain.J Neurosci Res. 2021 Oct;99(10):2610-2624. doi: 10.1002/jnr.24919. Epub 2021 Jul 26. J Neurosci Res. 2021. PMID: 34310750 Free PMC article.
References
-
- Leach CS. Biochemical and hematologic changes after short-term space flight. Microgravity Q. 1992;2:69–75. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

