Ciliomotor circuitry underlying whole-body coordination of ciliary activity in the Platynereis larva
- PMID: 28508746
- PMCID: PMC5531833
- DOI: 10.7554/eLife.26000
Ciliomotor circuitry underlying whole-body coordination of ciliary activity in the Platynereis larva
Abstract
Ciliated surfaces harbouring synchronously beating cilia can generate fluid flow or drive locomotion. In ciliary swimmers, ciliary beating, arrests, and changes in beat frequency are often coordinated across extended or discontinuous surfaces. To understand how such coordination is achieved, we studied the ciliated larvae of Platynereis dumerilii, a marine annelid. Platynereis larvae have segmental multiciliated cells that regularly display spontaneous coordinated ciliary arrests. We used whole-body connectomics, activity imaging, transgenesis, and neuron ablation to characterize the ciliomotor circuitry. We identified cholinergic, serotonergic, and catecholaminergic ciliomotor neurons. The synchronous rhythmic activation of cholinergic cells drives the coordinated arrests of all cilia. The serotonergic cells are active when cilia are beating. Serotonin inhibits the cholinergic rhythm, and increases ciliary beat frequency. Based on their connectivity and alternating activity, the catecholaminergic cells may generate the rhythm. The ciliomotor circuitry thus constitutes a stop-and-go pacemaker system for the whole-body coordination of ciliary locomotion.
Keywords: P. dumerilii; acetylcholine; catecholamines; ciliary nerve; connectomics; neuroscience; serotonin; zooplankton.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures






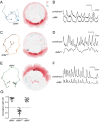

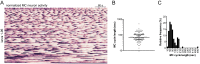

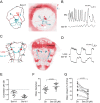
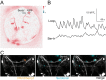
Comment in
-
A network for swimming.Elife. 2017 Jul 5;6:e28780. doi: 10.7554/eLife.28780. Elife. 2017. PMID: 28678696 Free PMC article.
Similar articles
-
Neuronal coordination of motile cilia in locomotion and feeding.Philos Trans R Soc Lond B Biol Sci. 2020 Feb 17;375(1792):20190165. doi: 10.1098/rstb.2019.0165. Epub 2019 Dec 30. Philos Trans R Soc Lond B Biol Sci. 2020. PMID: 31884921 Free PMC article. Review.
-
Mechanism of barotaxis in marine zooplankton.Elife. 2024 Sep 19;13:RP94306. doi: 10.7554/eLife.94306. Elife. 2024. PMID: 39298255 Free PMC article.
-
Neuropeptides regulate swimming depth of Platynereis larvae.Proc Natl Acad Sci U S A. 2011 Nov 15;108(46):E1174-83. doi: 10.1073/pnas.1109085108. Epub 2011 Oct 17. Proc Natl Acad Sci U S A. 2011. PMID: 22006315 Free PMC article.
-
Melatonin signaling controls circadian swimming behavior in marine zooplankton.Cell. 2014 Sep 25;159(1):46-57. doi: 10.1016/j.cell.2014.07.042. Cell. 2014. PMID: 25259919 Free PMC article.
-
Neuronal cell types in the annelid Platynereis dumerilii.Curr Opin Neurobiol. 2019 Jun;56:106-116. doi: 10.1016/j.conb.2018.12.008. Epub 2019 Jan 24. Curr Opin Neurobiol. 2019. PMID: 30685735 Review.
Cited by
-
Delegating Sex: Differential Gene Expression in Stolonizing Syllids Uncovers the Hormonal Control of Reproduction.Genome Biol Evol. 2019 Jan 1;11(1):295-318. doi: 10.1093/gbe/evy265. Genome Biol Evol. 2019. PMID: 30535381 Free PMC article.
-
Perspectives on Principles of Cellular Behavior from the Biophysics of Protists.Integr Comp Biol. 2023 Dec 29;63(6):1405-1421. doi: 10.1093/icb/icad106. Integr Comp Biol. 2023. PMID: 37496203 Free PMC article. Review.
-
The development of the adult nervous system in the annelid Owenia fusiformis.Neural Dev. 2024 Feb 21;19(1):3. doi: 10.1186/s13064-024-00180-8. Neural Dev. 2024. PMID: 38383501 Free PMC article.
-
Neural circuitry of a polycystin-mediated hydrodynamic startle response for predator avoidance.Elife. 2018 Dec 14;7:e36262. doi: 10.7554/eLife.36262. Elife. 2018. PMID: 30547885 Free PMC article.
-
Coordination of eukaryotic cilia and flagella.Essays Biochem. 2018 Dec 7;62(6):829-838. doi: 10.1042/EBC20180029. Print 2018 Dec 7. Essays Biochem. 2018. PMID: 30464007 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

