Ebola exposure, illness experience, and Ebola antibody prevalence in international responders to the West African Ebola epidemic 2014-2016: A cross-sectional study
- PMID: 28510604
- PMCID: PMC5433702
- DOI: 10.1371/journal.pmed.1002300
Ebola exposure, illness experience, and Ebola antibody prevalence in international responders to the West African Ebola epidemic 2014-2016: A cross-sectional study
Abstract
Background: Healthcare and other front-line workers are at particular risk of infection with Ebola virus (EBOV). Despite the large-scale deployment of international responders, few cases of Ebola virus disease have been diagnosed in this group. Since asymptomatic or pauci-symptomatic infection has been described, it is plausible that infections have occurred in healthcare workers but have escaped being diagnosed. We aimed to assess the prevalence of asymptomatic or pauci-symptomatic infection, and of exposure events, among returned responders to the West African Ebola epidemic 2014-2016.
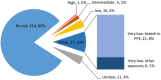
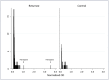
Methods and findings: We used snowball sampling to identify responders who had returned to the UK or Ireland, and used an online consent and questionnaire to determine their exposure to EBOV and their experience of illness. Oral fluid collection devices were sent and returned by post, and samples were tested using an EBOV IgG capture assay that detects IgG to Ebola glycoprotein. Blood was collected from returnees with reactive samples for further testing. Unexposed UK controls were also recruited. In all, 300 individuals consented, of whom 268 (89.3%) returned an oral fluid sample (OFS). The majority had worked in Sierra Leone in clinical, laboratory, research, and other roles. Fifty-three UK controls consented and provided samples using the same method. Of the returnees, 47 (17.5%) reported that they had had a possible EBOV exposure. Based on their free-text descriptions, using a published risk assessment method, we classified 43 (16%) as having had incidents with risk of Ebola transmission, including five intermediate-risk and one high-risk exposure. Of the returnees, 57 (21%) reported a febrile or diarrhoeal illness in West Africa or within 1 mo of return, of whom 40 (70%) were not tested at the time for EBOV infection. Of the 268 OFSs, 266 were unreactive. Two returnees, who did not experience an illness in West Africa or on return, had OFSs that were reactive on the EBOV IgG capture assay, with similar results on plasma. One individual had no further positive test results; the other had a positive result on a double-antigen bridging assay but not on a competitive assay or on an indirect EBOV IgG ELISA. All 53 controls had non-reactive OFSs. While the participants were not a random sample of returnees, the number participating was high.
Conclusions: This is the first study, to our knowledge, of the prevalence of EBOV infection in international responders. More than 99% had clear negative results. Sera from two individuals had discordant results on the different assays; both were negative on the competitive assay, suggesting that prior infection was unlikely. The finding that a significant proportion experienced "near miss" exposure events, and that most of those who experienced symptoms did not get tested for EBOV at the time, suggests a need to review and standardise protocols for the management of possible exposure to EBOV, and for the management of illness, across organisations that deploy staff to outbreaks.
Conflict of interest statement
I have read the journal's policy and the authors of this manuscript have the following competing interests: RT has received funding from the Wellcome Trust via the University of Liverpool, and has received non-financial support from NHSBT, as part of the Convalescent Plasma Study. All other authors have declared that no competing interests exist.
Figures
References
-
- World Health Organization (2016) WHO global health workforce statistics. Geneva: World Health Organization; http://www.who.int/hrh/statistics/hwfstats/. Accessed 21 September 2016.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

