Regionally compartmentalized resident memory T cells mediate naturally acquired protection against pneumococcal pneumonia
- PMID: 28513594
- PMCID: PMC5693795
- DOI: 10.1038/mi.2017.43
Regionally compartmentalized resident memory T cells mediate naturally acquired protection against pneumococcal pneumonia
Abstract
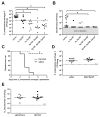
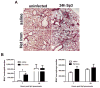
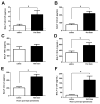
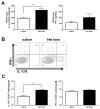
As children age, they become less susceptible to the diverse microbes causing pneumonia. These microbes are pathobionts that infect the respiratory tract multiple times during childhood, generating immunological memory. To elucidate mechanisms of such naturally acquired immune protection against pneumonia, we modeled a relevant immunological history in mice by infecting their airways with mismatched serotypes of Streptococcus pneumoniae (pneumococcus). Previous pneumococcal infections provided protection against a heterotypic, highly virulent pneumococcus, as evidenced by reduced bacterial burdens and long-term sterilizing immunity. This protection was diminished by depletion of CD4+ cells prior to the final infection. The resolution of previous pneumococcal infections seeded the lungs with CD4+ resident memory T (TRM) cells, which responded to heterotypic pneumococcus stimulation by producing multiple effector cytokines, particularly interleukin (IL)-17A. Following lobar pneumonias, IL-17-producing CD4+ TRM cells were confined to the previously infected lobe, rather than dispersed throughout the lower respiratory tract. Importantly, pneumonia protection also was confined to that immunologically experienced lobe. Thus regionally localized memory cells provide superior local tissue protection to that mediated by systemic or central memory immune defenses. We conclude that respiratory bacterial infections elicit CD4+ TRM cells that fill a local niche to optimize heterotypic protection of the affected tissue, preventing pneumonia.
Conflict of interest statement
Figures

Similar articles
-
Lung CD4+ resident memory T cells remodel epithelial responses to accelerate neutrophil recruitment during pneumonia.Mucosal Immunol. 2020 Mar;13(2):334-343. doi: 10.1038/s41385-019-0229-2. Epub 2019 Nov 20. Mucosal Immunol. 2020. PMID: 31748706 Free PMC article.
-
Generation of protective pneumococcal-specific nasal resident memory CD4+ T cells via parenteral immunization.Mucosal Immunol. 2020 Jan;13(1):172-182. doi: 10.1038/s41385-019-0218-5. Epub 2019 Oct 28. Mucosal Immunol. 2020. PMID: 31659300 Free PMC article.
-
B cells in the pneumococcus-infected lung are heterogeneous and require CD4+ T cell help including CD40L to become resident memory B cells.Front Immunol. 2024 Apr 23;15:1382638. doi: 10.3389/fimmu.2024.1382638. eCollection 2024. Front Immunol. 2024. PMID: 38715601 Free PMC article.
-
The Role of CD4+ Resident Memory T Cells in Local Immunity in the Mucosal Tissue - Protection Versus Pathology.Front Immunol. 2021 Apr 21;12:616309. doi: 10.3389/fimmu.2021.616309. eCollection 2021. Front Immunol. 2021. PMID: 33968018 Free PMC article. Review.
-
Inflammation and Pneumonia: Why Are Some More Susceptible than Others?Clin Chest Med. 2018 Dec;39(4):669-676. doi: 10.1016/j.ccm.2018.07.002. Clin Chest Med. 2018. PMID: 30390740 Free PMC article. Review.
Cited by
-
CD4 TRM Cells Following Infection and Immunization: Implications for More Effective Vaccine Design.Front Immunol. 2018 Aug 10;9:1860. doi: 10.3389/fimmu.2018.01860. eCollection 2018. Front Immunol. 2018. PMID: 30147701 Free PMC article. Review.
-
Evaluation of the Role of stat3 in Antibody and TH17-Mediated Responses to Pneumococcal Immunization and Infection by Use of a Mouse Model of Autosomal Dominant Hyper-IgE Syndrome.Infect Immun. 2018 Apr 23;86(5):e00024-18. doi: 10.1128/IAI.00024-18. Print 2018 May. Infect Immun. 2018. PMID: 29463618 Free PMC article.
-
Tissue-resident memory T cells trigger rapid exudation and local antibody accumulation.Mucosal Immunol. 2023 Feb;16(1):17-26. doi: 10.1016/j.mucimm.2022.11.004. Epub 2023 Jan 16. Mucosal Immunol. 2023. PMID: 36657662 Free PMC article.
-
Gearing up for battle: Harnessing adaptive T cell immunity against gram-negative pneumonia.Front Cell Infect Microbiol. 2022 Aug 18;12:934671. doi: 10.3389/fcimb.2022.934671. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 36061870 Free PMC article. Review.
-
Pneumonia recovery reprograms the alveolar macrophage pool.JCI Insight. 2020 Feb 27;5(4):e133042. doi: 10.1172/jci.insight.133042. JCI Insight. 2020. PMID: 31990682 Free PMC article.
References
-
- Bogaert D, De Groot R, Hermans PW. Streptococcus pneumoniae colonisation: the key to pneumococcal disease. Lancet Infect Dis. 2004;4(3):144–154. - PubMed
-
- Curns AT, Holman RC, Sejvar JJ, Owings MF, Schonberger LB. Infectious disease hospitalizations among older adults in the United States from 1990 through 2002. Arch Intern Med. 2005;165(21):2514–2520. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

