Lack of MTTP Activity in Pluripotent Stem Cell-Derived Hepatocytes and Cardiomyocytes Abolishes apoB Secretion and Increases Cell Stress
- PMID: 28514664
- PMCID: PMC5555078
- DOI: 10.1016/j.celrep.2017.04.064
Lack of MTTP Activity in Pluripotent Stem Cell-Derived Hepatocytes and Cardiomyocytes Abolishes apoB Secretion and Increases Cell Stress
Abstract
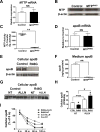
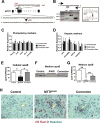
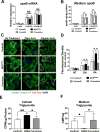
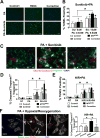
Abetalipoproteinemia (ABL) is an inherited disorder of lipoprotein metabolism resulting from mutations in microsomal triglyceride transfer protein (MTTP). In addition to expression in the liver and intestine, MTTP is expressed in cardiomyocytes, and cardiomyopathy has been reported in several ABL cases. Using induced pluripotent stem cells (iPSCs) generated from an ABL patient homozygous for a missense mutation (MTTPR46G), we show that human hepatocytes and cardiomyocytes exhibit defects associated with ABL disease, including loss of apolipoprotein B (apoB) secretion and intracellular accumulation of lipids. MTTPR46G iPSC-derived cardiomyocytes failed to secrete apoB, accumulated intracellular lipids, and displayed increased cell death, suggesting intrinsic defects in lipid metabolism due to loss of MTTP function. Importantly, these phenotypes were reversed after the correction of the MTTPR46G mutation by CRISPR/Cas9 gene editing. Together, these data reveal clear cellular defects in iPSC-derived hepatocytes and cardiomyocytes lacking MTTP activity, including a cardiomyocyte-specific regulated stress response to elevated lipids.
Keywords: abetaliproteinemia; apoB; cardiac stress; iPSC-derived hepatocytes and cardiomyocytes; induced pluripotent stem cells; lipid accumulation.
Copyright © 2017 The Authors. Published by Elsevier Inc. All rights reserved.
Figures


References
-
- Al-Shali K, Wang J, Rosen F, Hegele RA. Ileal adenocarcinoma in a mild phenotype of abetalipoproteinemia. Clin Genet. 2003;63:135–138. - PubMed
-
- Athar H, Iqbal J, Jiang XC, Hussain MM. A simple, rapid, and sensitive fluorescence assay for microsomal triglyceride transfer protein. J Lipid Res. 2004;45:764–772. - PubMed
-
- Berriot-Varoqueaux N, Aggerbeck LP, Samson-Bouma M, Wetterau JR. The role of the microsomal triglygeride transfer protein in abetalipoproteinemia. Annu Rev Nutr. 2000;20:663–697. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

