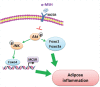
αMSH inhibits adipose inflammation via reducing FoxOs transcription and blocking Akt/JNK pathway in mice
- PMID: 28514752
- PMCID: PMC5564594
- DOI: 10.18632/oncotarget.17465
αMSH inhibits adipose inflammation via reducing FoxOs transcription and blocking Akt/JNK pathway in mice
Abstract
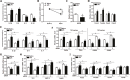
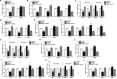
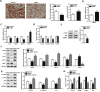
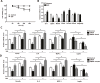
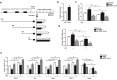
Alpha melanocyte stimulating hormone (αMSH) abates inflammation in multiple tissues, while Forkhead box proteins O (FoxOs) stimulate inflammatory cascade. However, the relationship between αMSH and FoxOs in adipose inflammation remains unclear. In this study, we used LPS-induced inflammation model, attempted to interpret the function of αMSH in inflammation and the interactions with FoxOs. Results indicated that upon inflammatory situation, the secretion of αMSH and the expression of its receptor MC5R were greatly decreased, but FoxOs expressions were elevated. After the treatment with αMSH, LPS-induced adipose inflammation together with FoxOs expressions was significantly reduced. Conversely, when Foxo1, Foxo3a or Foxo4 overexpressed in αMSH treated inflammatory mouse model, all the anti-inflammatory impacts of αMSH were found disappeared. We further studied the mechanisms by which αMSH exerts its anti-inflammatory impacts and how FoxOs reverse αMSH's function. Foxo4 was found as a negative regulator for MC5R transcription in αMSH inhibited inflammation. Moreover, a negative role was found of αMSH in regulating both Akt and JNK signal pathways by observing the enhanced the anti-inflammatory impacts of pathway-specific inhibitors with αMSH treatment. Our findings demonstrate αMSH plays a key role in the prevention of adipose inflammation and inflammatory diseases by down-regulating Akt/JNK signal pathway and negatively interacting with FoxOs, which brings up αMSH as a novel candidate factor in the adipose anti-inflammation process in obesity.
Keywords: Akt/JNK; FoxOs; adipose; inflammation; αMSH.
Conflict of interest statement
The authors declare no conflicts of interest associated with this manuscript.
Figures
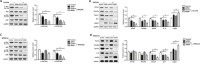
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

