Patient-centered primary care for adults at high risk for AUDs: the Choosing Healthier Drinking Options In primary CarE (CHOICE) trial
- PMID: 28514963
- PMCID: PMC5436432
- DOI: 10.1186/s13722-017-0080-2
Patient-centered primary care for adults at high risk for AUDs: the Choosing Healthier Drinking Options In primary CarE (CHOICE) trial
Abstract
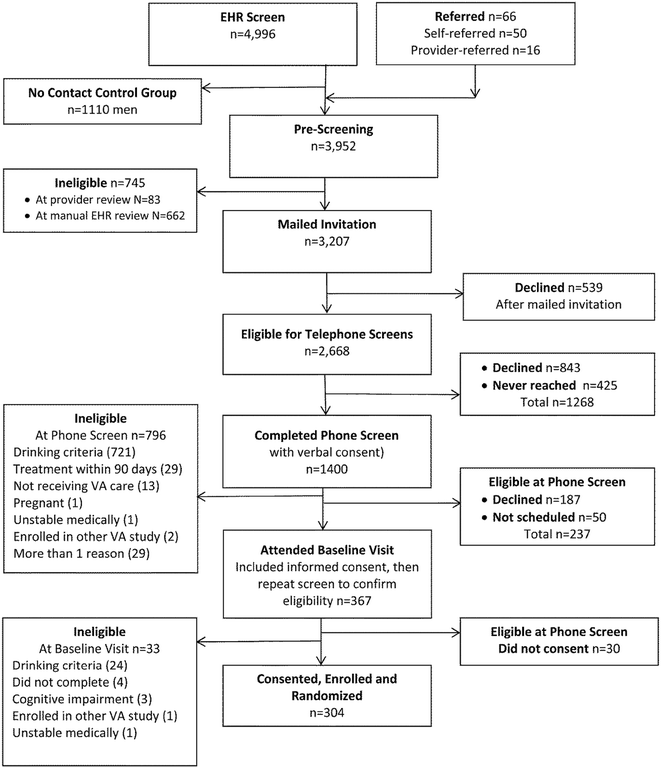
Background: Most patients with alcohol use disorders (AUDs) never receive alcohol treatment, and experts have recommended management of AUDs in primary care. The Choosing Healthier Drinking Options In primary CarE (CHOICE) trial was a randomized controlled effectiveness trial of a novel intervention for primary care patients at high risk for AUDs. This report describes the conceptual and scientific foundation of the CHOICE model of care, critical elements of the CHOICE trial design consistent with the Template for Intervention Description and Replication (TIDieR), results of recruitment, and baseline characteristics of the enrolled sample.
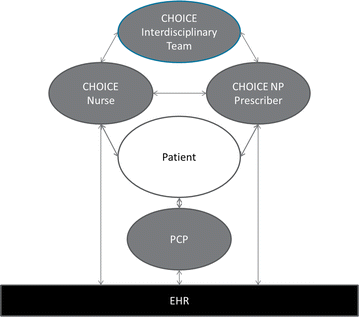
Methods: The CHOICE intervention is a multi-contact, extended counseling intervention, based on the Chronic Care Model, shared decision-making, motivational interviewing, and evidence-based options for managing AUDs, designed to be practical in primary care. Outpatients who received care at 3 Veterans Affairs primary care sites in the Pacific Northwest and reported frequent heavy drinking (≥4 drinks/day for women; ≥5 for men) were recruited (2011-2014) into a trial in which half of the participants would be offered additional alcohol-related care from a nurse. CHOICE nurses offered 12 months of patient-centered care, including proactive outreach and engagement, repeated brief motivational interventions, monitoring with and without alcohol biomarkers, medications for AUDs, and/or specialty alcohol treatment as appropriate and per patient preference. A CHOICE nurse practitioner was available to prescribe medications for AUDs.
Results: A total of 304 patients consented to participate in the CHOICE trial. Among consenting participants, 90% were men, the mean age was 51 (range 22-75), and most met DSM-IV criteria for alcohol abuse (14%) or dependence (59%). Many participants also screened positive for tobacco use (44%), depression (45%), anxiety disorders (30-41%) and non-tobacco drug use disorders (19%). At baseline, participants had a median AUDIT score of 18 [Interquartile range (IQR) 14-24] and a median readiness to change drinking score of 5 (IQR 2.75-6.25) on a 1-10 Likert scale.
Conclusion: The CHOICE trial tested a patient-centered intervention for AUDs and recruited primary care patients at high risk for AUDs, with a spectrum of severity, co-morbidity, and readiness to change drinking. Trial registration The trial is registered at clinicaltrial.gov (NCT01400581).
Keywords: Alcohol use disorder; Brief interventions; Care management; Chronic Care Model; Intervention; Medical management; Patient-centered care; Primary care; Shared decision making; Veterans.
Figures
Similar articles
-
Barriers and facilitators to changing drinking and receiving alcohol-related care: Interviews with Veterans Health Administration primary care patients who indicated interest but did not enroll in an alcohol care management intervention trial.Subst Abus. 2022;43(1):1197-1206. doi: 10.1080/08897077.2022.2074602. Subst Abus. 2022. PMID: 35657656 Free PMC article.
-
Alcohol-Related Nurse Care Management in Primary Care: A Randomized Clinical Trial.JAMA Intern Med. 2018 May 1;178(5):613-621. doi: 10.1001/jamainternmed.2018.0388. JAMA Intern Med. 2018. PMID: 29582088 Free PMC article. Clinical Trial.
-
Comparison of DSM-IV and DSM-5 criteria for alcohol use disorders in VA primary care patients with frequent heavy drinking enrolled in a trial.Addict Sci Clin Pract. 2017 Jul 18;12(1):17. doi: 10.1186/s13722-017-0082-0. Addict Sci Clin Pract. 2017. PMID: 28716049 Free PMC article.
-
Alcoholics Anonymous and other 12-step programs for alcohol use disorder.Cochrane Database Syst Rev. 2020 Mar 11;3(3):CD012880. doi: 10.1002/14651858.CD012880.pub2. Cochrane Database Syst Rev. 2020. PMID: 32159228 Free PMC article.
-
The role of screening, brief intervention, and referral to treatment in the perinatal period.Am J Obstet Gynecol. 2016 Nov;215(5):539-547. doi: 10.1016/j.ajog.2016.06.038. Epub 2016 Jul 1. Am J Obstet Gynecol. 2016. PMID: 27373599 Free PMC article. Review.
Cited by
-
Treating alcohol use disorders in primary care - a qualitative evaluation of a new innovation: the 15-method.Scand J Prim Health Care. 2021 Mar;39(1):51-59. doi: 10.1080/02813432.2021.1882079. Epub 2021 Feb 15. Scand J Prim Health Care. 2021. PMID: 33586596 Free PMC article. Clinical Trial.
-
Usability and Acceptability of a Mobile App for the Self-Management of Alcohol Misuse Among Veterans (Step Away): Pilot Cohort Study.JMIR Mhealth Uhealth. 2021 Apr 8;9(4):e25927. doi: 10.2196/25927. JMIR Mhealth Uhealth. 2021. PMID: 33830064 Free PMC article.
-
Strategies to increase implementation of pharmacotherapy for alcohol use disorders: a structured review of care delivery and implementation interventions.Addict Sci Clin Pract. 2019 Feb 12;14(1):6. doi: 10.1186/s13722-019-0134-8. Addict Sci Clin Pract. 2019. PMID: 30744686 Free PMC article. Review.
-
Barriers and facilitators to changing drinking and receiving alcohol-related care: Interviews with Veterans Health Administration primary care patients who indicated interest but did not enroll in an alcohol care management intervention trial.Subst Abus. 2022;43(1):1197-1206. doi: 10.1080/08897077.2022.2074602. Subst Abus. 2022. PMID: 35657656 Free PMC article.
-
Integration of pharmacotherapy for alcohol use disorder treatment in primary care settings: A scoping review.J Subst Abuse Treat. 2023 Jan;144:108919. doi: 10.1016/j.jsat.2022.108919. Epub 2022 Oct 28. J Subst Abuse Treat. 2023. PMID: 36332528 Free PMC article.
References
-
- Grant BF, Goldstein RB, Saha TD, Chou SP, Jung J, Zhang H, Pickering RP, Ruan WJ, Smith SM, Huang B, Hasin DS. Epidemiology of DSM-5 alcohol use disorder: results from the National Epidemiologic Survey on alcohol and related conditions III. JAMA Psychiatry. 2015;72:757–766. doi: 10.1001/jamapsychiatry.2015.0584. - DOI - PMC - PubMed
-
- Substance Abuse and Mental Health Services Administration: Results from the 2013 National Survey on Drug Use and Health: Summary of National Findings. In: NSDUH Series H-48, HHS Publication No. (SMA) 14-4863. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2014.
-
- Institute of Medicine . Broadening the base of treatment for alcohol problems: a report of the committee for the study of treatment and rehabilitation for alcoholism. Washington: National Academy Press; 1990.
-
- Institute of Medicine . Improving the quality of health care for mental and substance-use conditions: quality Chasm series. Washington: National Academies Press; 2006. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous