Aberrant Network Activity in Schizophrenia
- PMID: 28515010
- PMCID: PMC5523137
- DOI: 10.1016/j.tins.2017.04.003
Aberrant Network Activity in Schizophrenia
Abstract
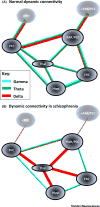
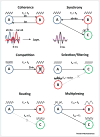
Brain dynamic changes associated with schizophrenia are largely equivocal, with interpretation complicated by many factors, such as the presence of therapeutic agents and the complex nature of the syndrome itself. Evidence for a brain-wide change in individual network oscillations, shared by all patients, is largely equivocal, but stronger for lower (delta) than for higher (gamma) bands. However, region-specific changes in rhythms across multiple, interdependent, nested frequencies may correlate better with pathology. Changes in synaptic excitation and inhibition in schizophrenia disrupt delta rhythm-mediated cortico-cortical communication, while enhancing thalamocortical communication in this frequency band. The contrasting relationships between delta and higher frequencies in thalamus and cortex generate frequency mismatches in inter-regional connectivity, leading to a disruption in temporal communication between higher-order brain regions associated with mental time travel.
Keywords: cross-frequency coupling; default mode network; delta rhythm; gamma rhythm; thalamocortical communication; theta rhythm.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Figures



References
-
- Garrity AG. Aberrant “default mode” functional connectivity in schizophrenia. Am. J. Psychiatry. 2007;164:450–457. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

