ω-3 and ω-6 long-chain PUFAs and their enzymatic metabolites in neovascular eye diseases
- PMID: 28515072
- PMCID: PMC5486202
- DOI: 10.3945/ajcn.117.153825
ω-3 and ω-6 long-chain PUFAs and their enzymatic metabolites in neovascular eye diseases
Abstract
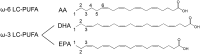
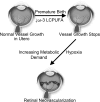
Neovascular eye diseases, including retinopathy of prematurity, diabetic retinopathy, and age-related macular degeneration, threaten the visual health of children and adults. Current treatment options, including anti-vascular endothelial growth factor therapy and laser retinal photocoagulation, have limitations and are associated with adverse effects; therefore, the identification of additional therapies is highly desirable. Both clinical and experimental studies show that dietary ω-3 (n-3) long-chain polyunsaturated fatty acids (LC-PUFAs) reduce retinal and choroidal angiogenesis. The ω-3 LC-PUFA metabolites from 2 groups of enzymes, cyclooxygenases and lipoxygenases, inhibit [and the ω-6 (n-6) LC-PUFA metabolites promote] inflammation and angiogenesis. However, both of the ω-3 and the ω-6 lipid products of cytochrome P450 oxidase 2C promote neovascularization in both the retina and choroid, which suggests that inhibition of this pathway might be beneficial. This review summarizes our current understanding of the roles of ω-3 and ω-6 LC-PUFAs and their enzymatic metabolites in neovascular eye diseases.
Keywords: age-related macular degeneration; diabetic retinopathy; lipid metabolism; long-chain polyunsaturated fatty acid; retinopathy of prematurity.
© 2017 American Society for Nutrition.
Figures



References
-
- Gibson DM. Diabetic retinopathy and age-related macular degeneration in the U.S. Am J Prev Med 2012;43:48–54. - PubMed
-
- Comparison of Age-related Macular Degeneration Treatments Trials Research Group; Maguire MG, Martin DF, Ying GS, Jaffe GJ, Daniel E, Grunwald JE, Toth CA, Ferris FL III, Fine SL. Five-year outcomes with anti-vascular endothelial growth factor treatment of neovascular age-related macular degeneration: the comparison of age-related macular degeneration treatments trials. Ophthalmology 2016;123:1751–61. - PMC - PubMed
-
- Cheung N, Lam DS, Wong TY. Anti-vascular endothelial growth factor treatment for eye diseases. BMJ 2012;344:e2970. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

