Polymorphisms in methotrexate transporters and their relationship to plasma methotrexate levels, toxicity of high-dose methotrexate, and outcome of pediatric acute lymphoblastic leukemia
- PMID: 28525903
- PMCID: PMC5514947
- DOI: 10.18632/oncotarget.17781
Polymorphisms in methotrexate transporters and their relationship to plasma methotrexate levels, toxicity of high-dose methotrexate, and outcome of pediatric acute lymphoblastic leukemia
Abstract
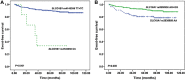
High-dose methotrexate (HDMTX) plays an important role in the treatment of acute lymphoblastic leukemia (ALL) although there is great inter-patient variability in the efficacy and toxicity of MTX. The relationship between polymorphisms in genes encoding MTX transporters and MTX response is controversial. In the present study, 322 Chinese children with standard- and intermediate-risk ALL were genotyped for 12 polymorphisms. SLCO1B1 rs10841753 showed a significant association with plasma MTX levels at 48 h (P = 0.017). Patients who had the ABCB1 rs1128503 C allele had longer duration of hospitalization than did those with the TT genotype (P = 0.006). No association was found between oral mucositis and any polymorphism. Long-term outcome was worse in patients with the SLCO1B1 rs4149056 CC genotype than in patients with TT or TC (5-year event-free survival [EFS] 33.3 ± 19.2% vs. 90.5 ± 1.7%, P < 0.001), and was worse in patients with the SCL19A1 rs2838958 AA genotype than in patients with AG or GG (5-year EFS 78.5 ± 4.6% vs. 92.2 ± 1.8%, P = 0.008). Multiple Cox regression analyses revealed associations of minimal residual disease (MRD) at day 33 (hazard ratio 3.458; P = 0.002), MRD at day 78 (hazard ratio 6.330; P = 0.001), SLCO1B1 rs4149056 (hazard ratio 12.242; P < 0.001), and SCL19A1 rs2838958 (hazard ratio 2.324; P = 0.019) with EFS. Our findings show that polymorphisms in genes encoding MTX transporters substantially influence the kinetics and response to HDMTX therapy in childhood ALL.
Keywords: SLC19A1; SLCO1B1; acute lymphoblastic leukemia; methotrexate; polymorphism.
Conflict of interest statement
None.
Figures
Similar articles
-
[SLCO1B1c. 521T>C gene polymorphisms are associated with high-dose methotrexate pharmacokinetics and clinical outcome of pediatric acute lymphoblastic leukemia].Zhonghua Er Ke Za Zhi. 2014 Oct;52(10):770-6. Zhonghua Er Ke Za Zhi. 2014. PMID: 25537545 Chinese.
-
Impact of SLCO1B1 521T > C variant on leucovorin rescue and risk of relapse in childhood acute lymphoblastic leukemia treated with high-dose methotrexate.Pediatr Blood Cancer. 2014 Dec;61(12):2203-7. doi: 10.1002/pbc.25191. Epub 2014 Aug 17. Pediatr Blood Cancer. 2014. PMID: 25130190
-
Polymorphisms of the SLCO1B1 gene predict methotrexate-related toxicity in childhood acute lymphoblastic leukemia.Pediatr Blood Cancer. 2011 Oct;57(4):612-9. doi: 10.1002/pbc.23074. Epub 2011 Mar 8. Pediatr Blood Cancer. 2011. PMID: 21387541
-
Methotrexate Disposition in Pediatric Patients with Acute Lymphoblastic Leukemia: What Have We Learnt From the Genetic Variants of Drug Transporters.Curr Pharm Des. 2019;25(6):627-634. doi: 10.2174/1381612825666190329141003. Curr Pharm Des. 2019. PMID: 30931851 Review.
-
Association between ABCB1 C3435 T polymorphism- and methotrexate-related toxicity in pediatric acute lymphoblastic leukemia: a meta-analysis.Hematology. 2025 Dec;30(1):2469373. doi: 10.1080/16078454.2025.2469373. Epub 2025 Mar 3. Hematology. 2025. PMID: 40029037 Review.
Cited by
-
Systematic review: genetic polymorphisms in the pharmacokinetics of high-dose methotrexate in pediatric acute lymphoblastic leukemia patients.Cancer Chemother Pharmacol. 2024 Aug;94(2):141-155. doi: 10.1007/s00280-024-04694-0. Epub 2024 Jul 13. Cancer Chemother Pharmacol. 2024. PMID: 39002021
-
Promising genes and variants to reduce chemotherapy adverse effects in acute lymphoblastic leukemia.Transl Oncol. 2021 Jan;14(1):100978. doi: 10.1016/j.tranon.2020.100978. Epub 2020 Dec 5. Transl Oncol. 2021. PMID: 33290991 Free PMC article. Review.
-
Population Pharmacokinetics of High-Dose Methotrexate in Chinese Pediatric Patients With Acute Lymphoblastic Leukemia.Front Pharmacol. 2021 Jul 13;12:701452. doi: 10.3389/fphar.2021.701452. eCollection 2021. Front Pharmacol. 2021. PMID: 34326772 Free PMC article.
-
Systematic Review of Pharmacogenetic Factors That Influence High-Dose Methotrexate Pharmacokinetics in Pediatric Malignancies.Cancers (Basel). 2021 Jun 7;13(11):2837. doi: 10.3390/cancers13112837. Cancers (Basel). 2021. PMID: 34200242 Free PMC article. Review.
-
Polymorphisms within methotrexate pathway genes: Relationship between plasma methotrexate levels, toxicity experienced and outcome in pediatric acute lymphoblastic leukemia.Iran J Basic Med Sci. 2020 Jun;23(6):800-809. doi: 10.22038/ijbms.2020.41754.9858. Iran J Basic Med Sci. 2020. PMID: 32695297 Free PMC article.
References
-
- Liu SG, Li ZG, Cui L, Gao C, Li WJ, Zhao XX. Effects of methylenetetrahydrofolate reductase gene polymorphisms on toxicities during consolidation therapy in pediatric acute lymphoblastic leukemia in a Chinese population. Leuk Lymphoma. 2011;52:1030–1040. - PubMed
-
- Csordas K, Lautner-Csorba O, Semsei AF, Harnos A, Hegyi M, Erdelyi DJ, Eipel OT, Szalai C, Kovacs GT. Associations of novel genetic variations in the folate-related and ARID5B genes with the pharmacokinetics and toxicity of high-dose methotrexate in paediatric acute lymphoblastic leukaemia. Br J Haematol. 2014;166:410–420. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

