Ubiquitin-Dependent Regulation of Stem Cell Biology
- PMID: 28528988
- PMCID: PMC5643009
- DOI: 10.1016/j.tcb.2017.04.002
Ubiquitin-Dependent Regulation of Stem Cell Biology
Abstract
The growth of a metazoan body relies on a series of highly coordinated cell-fate decisions by stem cells which can undergo self-renewal, reversibly enter a quiescent state, or terminally commit to a cell specification program. To guide their decisions, stem cells make frequent use of ubiquitylation, a post-translational modification that can affect the activity, interaction landscape, or stability of stem cell proteins. In this review we discuss novel findings that have provided insight into ubiquitin-dependent mechanisms of stem cell control and revealed how an essential and highly conserved protein modification can shape metazoan development.
Keywords: E3 ligase; development; differentiation; stem cell; ubiquitin.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Figures


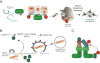
References
-
- Thomson JA, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282(5391):1145–7. - PubMed
-
- Becker AJ, et al. Cytological demonstration of the clonal nature of spleen colonies derived from transplanted mouse marrow cells. Nature. 1963;197:452–4. - PubMed
-
- Leblond CP, Walker BE. Renewal of cell populations. Physiol Rev. 1956;36(2):255–76. - PubMed
-
- Yau R, Rape M. The increasing complexity of the ubiquitin code. Nat Cell Biol. 2016;18(6):579–86. - PubMed
-
- Deshaies RJ, Joazeiro CA. RING domain E3 ubiquitin ligases. Annu Rev Biochem. 2009;78:399–434. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

