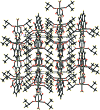
Crystal structure of aqua- trans-bis-(dimethyl sulfoxide-κ O)(pyridine-2,6-di-carboxyl-ato-κ3O2, N, O6)nickel(II)
- PMID: 28529797
- PMCID: PMC5418805
- DOI: 10.1107/S2056989017006090
Crystal structure of aqua- trans-bis-(dimethyl sulfoxide-κ O)(pyridine-2,6-di-carboxyl-ato-κ3O2, N, O6)nickel(II)
Abstract
In the title complex, [Ni(C7H3NO4)(C2H6OS)2(H2O)], the NiII cation is situated on a twofold rotation axis and exhibits a distorted octa-hedral NO5 coordination environment defined by a tridentate pyridine-2,6-di-carb-oxy-lic acid dianion (dpa2-), two dimethyl sulfoxide (DMSO) mol-ecules, and a water mol-ecule. In the crystal, the complex mol-ecules are linked by O-H⋯O and C-H⋯O hydrogen bonds into a three-dimensional network whereby DMSO mol-ecules from neighboring complexes overlap to form layers parallel to (001), alternating with layers of NiII-dpa2- moieties. The title complex is isotypic with its cobalt(II) analogue.
Keywords: crystal structure; dimethyl sulfoxide; hydrogen bonding; nickel(II); pyridine-2,6-dicarboxylic acid dianion.
Figures
Similar articles
-
Crystal structure of catena-poly[[(dimethyl sulfoxide-κO)(pyridine-2,6-di-carboxyl-ato-κ(3) O,N,O')nickel(II)]-μ-pyrazine-κ(2) N:N'].Acta Crystallogr E Crystallogr Commun. 2016 Apr 29;72(Pt 5):768-71. doi: 10.1107/S2056989016007064. eCollection 2016 May 1. Acta Crystallogr E Crystallogr Commun. 2016. PMID: 27308038 Free PMC article.
-
Crystal structure of trans-di-aqua-(1,4,8,11-tetra-aza-undeca-ne)nickel(II) bis-(pyridine-2,6-di-carboxyl-ato)nickel(II).Acta Crystallogr E Crystallogr Commun. 2021 Oct 29;77(Pt 11):1175-1179. doi: 10.1107/S2056989021011178. eCollection 2021 Nov 1. Acta Crystallogr E Crystallogr Commun. 2021. PMID: 34868658 Free PMC article.
-
Isotypic MnII and FeII binuclear complexes of the ligand 5,6-bis-(pyridin-2-yl)-pyrazine-2,3-di-carb-oxy-lic acid.Acta Crystallogr E Crystallogr Commun. 2016 Sep 9;72(Pt 10):1412-1416. doi: 10.1107/S2056989016014055. eCollection 2016 Oct 1. Acta Crystallogr E Crystallogr Commun. 2016. PMID: 27746931 Free PMC article.
-
Crystal structure of tetra-aqua-bis(3,5-di-amino-4H-1,2,4-triazol-1-ium)cobalt(II) bis-[bis-(pyridine-2,6-di-carboxyl-ato)cobaltate(II)] dihydrate.Acta Crystallogr E Crystallogr Commun. 2015 May 30;71(Pt 6):m139-40. doi: 10.1107/S2056989015010014. eCollection 2015 Jun 1. Acta Crystallogr E Crystallogr Commun. 2015. PMID: 26090171 Free PMC article.
-
Synthesis and structural characterization of the dichloride complex formed by carb-oxy-functionalized Cu(di-aza-cyclam)2+ cation and its heterometallic coordination polymer with CdCl2.Acta Crystallogr E Crystallogr Commun. 2025 May 2;81(Pt 6):458-463. doi: 10.1107/S2056989025003792. eCollection 2025 Jun 1. Acta Crystallogr E Crystallogr Commun. 2025. PMID: 40487698 Free PMC article.
References
-
- Bruker (2014). APEX2, SAINT and SADABS. Bruker Inc., Madison, Wisconsin, USA.
-
- Du, M., Li, C. P., Liu, C. S. & Fang, S. M. (2013). Coord. Chem. Rev. 257, 1282–1305.
-
- Felloni, M., Blake, A. J., Hubberstey, P., Teat, S. J., Wilson, C. & Schröder, M. (2010). CrystEngComm, 12, 1576–1589.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous


 − z.
− z.