Metabolic basis to Sherpa altitude adaptation
- PMID: 28533386
- PMCID: PMC5474778
- DOI: 10.1073/pnas.1700527114
Metabolic basis to Sherpa altitude adaptation
Abstract
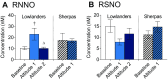
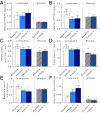
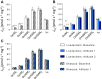
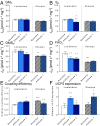
The Himalayan Sherpas, a human population of Tibetan descent, are highly adapted to life in the hypobaric hypoxia of high altitude. Mechanisms involving enhanced tissue oxygen delivery in comparison to Lowlander populations have been postulated to play a role in such adaptation. Whether differences in tissue oxygen utilization (i.e., metabolic adaptation) underpin this adaptation is not known, however. We sought to address this issue, applying parallel molecular, biochemical, physiological, and genetic approaches to the study of Sherpas and native Lowlanders, studied before and during exposure to hypobaric hypoxia on a gradual ascent to Mount Everest Base Camp (5,300 m). Compared with Lowlanders, Sherpas demonstrated a lower capacity for fatty acid oxidation in skeletal muscle biopsies, along with enhanced efficiency of oxygen utilization, improved muscle energetics, and protection against oxidative stress. This adaptation appeared to be related, in part, to a putatively advantageous allele for the peroxisome proliferator-activated receptor A (PPARA) gene, which was enriched in the Sherpas compared with the Lowlanders. Our findings suggest that metabolic adaptations underpin human evolution to life at high altitude, and could have an impact upon our understanding of human diseases in which hypoxia is a feature.
Keywords: altitude; hypoxia; metabolism; mitochondria; skeletal muscle.
Conflict of interest statement
Conflict of interest statement: E.G. is Chief Executive Officer and V.L. is Chief Operating Officer of Oroboros Instruments.
Figures


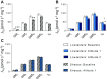
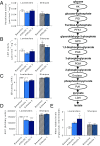

References
-
- Willam C, Nicholls LG, Ratcliffe PJ, Pugh CW, Maxwell PH. The prolyl hydroxylase enzymes that act as oxygen sensors regulating destruction of hypoxia-inducible factor alpha. Adv Enzyme Regul. 2004;44:75–92. - PubMed
-
- Aldenderfer M. Peopling the Tibetan plateau: Insights from archaeology. High Alt Med Biol. 2011;12:141–147. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- BB/F016581/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- FS/09/050/BHF_/British Heart Foundation/United Kingdom
- MR/P01836X/1/MRC_/Medical Research Council/United Kingdom
- MR/P011705/1/MRC_/Medical Research Council/United Kingdom
- MC_UP_A090_1006/MRC_/Medical Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources

