In Vitro Radiosensitizing Effects of Temozolomide on U87MG Cell Lines of Human Glioblastoma Multiforme
- PMID: 28533574
- PMCID: PMC5429494
In Vitro Radiosensitizing Effects of Temozolomide on U87MG Cell Lines of Human Glioblastoma Multiforme
Abstract
Background: Glioma is the most common primary brain tumor with poor prognosis. Temozolomide (TMZ) has been used with irradiation (IR) to treat gliomas. The aim of the present study was to evaluate the cytotoxic and radiosensitizing effect of TMZ when combined with high-dose and high-dose rate of gamma irradiation in vitro.
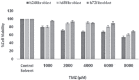
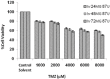
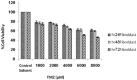
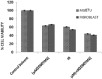
Methods: Two 'U87MG' cell lines and skin fibroblast were cultured and assigned to five groups for 24, 48, and 72 hours. The groups were namely, TMZ group (2000 μM/L), IR group (5 Gy), TMZ plus IR group, control group, and control solvent group. MTT assay was applied to evaluate cell viability. Data were analyzed with SPSS 21.0 software using one-way ANOVA and Kruskal-Wallis test. P<0.05 were considered statistically significant.
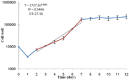
Results: The slope of growth curve U87MG cells in semi-logarithmic scale was equal to 27.36±0.89 hours. The viability of cells was determined for different TMZ and IR treatment groups. In terms of cell viability, there were no significant differences between the control and control solvent groups (P=0.35) and between treated group by IR (5 Gy) alone and TMZ (2000 µM/ml) alone (P=0.15). Data obtained for the cell viability of combined TMZ plus IR in both cell lines compared to TMZ or IR treated group alone showed a significant difference (P=0.002).
Conclusion: The evaluation of cells viability showed that TMZ in combination with IR on glioma cells led to a significant radiosensitivity compared to IR alone.
Keywords: Glioblastoma; Radiation-sensitizing agents; Radiotherapy; Temozolomide.
Figures
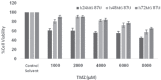
Similar articles
-
Radiosensitization of Glioma Cells by Temozolomide (TMZ): A Colony Formation Assay.J Biomed Phys Eng. 2022 Feb 1;12(1):43-50. doi: 10.31661/jbpe.v0i0.1223. eCollection 2022 Feb. J Biomed Phys Eng. 2022. PMID: 35155292 Free PMC article.
-
Radiosensitizing properties of dual-functionalized carbon nanostructures loaded with temozolomide.Beilstein J Nanotechnol. 2025 Feb 19;16:229-251. doi: 10.3762/bjnano.16.18. eCollection 2025. Beilstein J Nanotechnol. 2025. PMID: 39995757 Free PMC article.
-
Differential radiosensitizing potential of temozolomide in MGMT promoter methylated glioblastoma multiforme cell lines.Int J Radiat Oncol Biol Phys. 2007 Nov 15;69(4):1246-53. doi: 10.1016/j.ijrobp.2007.07.2366. Int J Radiat Oncol Biol Phys. 2007. PMID: 17967314
-
Combined treatment of Nimotuzumab and rapamycin is effective against temozolomide-resistant human gliomas regardless of the EGFR mutation status.BMC Cancer. 2015 Apr 11;15:255. doi: 10.1186/s12885-015-1191-3. BMC Cancer. 2015. PMID: 25886314 Free PMC article.
-
Suppression of the Eag1 potassium channel sensitizes glioblastoma cells to injury caused by temozolomide.Oncol Lett. 2016 Oct;12(4):2581-2589. doi: 10.3892/ol.2016.4992. Epub 2016 Aug 10. Oncol Lett. 2016. PMID: 27698831 Free PMC article.
Cited by
-
Radiosensitization of Glioma Cells by Temozolomide (TMZ): A Colony Formation Assay.J Biomed Phys Eng. 2022 Feb 1;12(1):43-50. doi: 10.31661/jbpe.v0i0.1223. eCollection 2022 Feb. J Biomed Phys Eng. 2022. PMID: 35155292 Free PMC article.
-
SLNP-based CDK4- targeted nanotherapy against glioblastoma.Front Oncol. 2024 Nov 22;14:1455816. doi: 10.3389/fonc.2024.1455816. eCollection 2024. Front Oncol. 2024. PMID: 39650055 Free PMC article.
-
Lower all-cause mortality rates in patients harboring pituitary carcinoma following the introduction of temozolomide.Endocrine. 2019 Aug;65(2):393-398. doi: 10.1007/s12020-019-01996-9. Epub 2019 Jul 2. Endocrine. 2019. PMID: 31267325
-
Radiosensitizing properties of dual-functionalized carbon nanostructures loaded with temozolomide.Beilstein J Nanotechnol. 2025 Feb 19;16:229-251. doi: 10.3762/bjnano.16.18. eCollection 2025. Beilstein J Nanotechnol. 2025. PMID: 39995757 Free PMC article.
-
Cobaltabis(dicarbollide) ([o-COSAN]-) as Multifunctional Chemotherapeutics: A Prospective Application in Boron Neutron Capture Therapy (BNCT) for Glioblastoma.Cancers (Basel). 2021 Dec 19;13(24):6367. doi: 10.3390/cancers13246367. Cancers (Basel). 2021. PMID: 34944987 Free PMC article.
References
-
- Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P, et al. An integrated genomic analysis of human glioblastoma multiforme. Science. 2008;321:1807–12. doi: 10.1126/science.1164382. [ PMC Free Article] - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
