Sodium glucose cotransporter 2 in mesangial cells and retinal pericytes and its implications for diabetic nephropathy and retinopathy
- PMID: 28535208
- PMCID: PMC5881757
- DOI: 10.1093/glycob/cwx047
Sodium glucose cotransporter 2 in mesangial cells and retinal pericytes and its implications for diabetic nephropathy and retinopathy
Abstract
Retinopathy and nephropathy are life-threatening diabetic complications that decrease patient quality of life. Although the mechanisms underlying these conditions have been extensively studied, they remain unknown. Recent reports have demonstrated the presence of sodium glucose cotransporter 2 (SGLT2) in retinal pericytes and mesangial cells. Hyperglycemia results in functional and morphological changes in these cells, but these effects are attenuated by phlorizin, a nonselective SGLT inhibitor. Based on these findings, we hypothesized that SGLT2 plays a pivotal role in the development of diabetic nephropathy and retinopathy and that SGLT2 inhibitors may directly protect against these complications.
Keywords: diabetic nephropathy; diabetic retinopathy; mesangial cell; retinal pericytes; sodium glucose cotransporter 2.
© The Author 2017. Published by Oxford University Press. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures
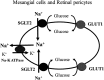
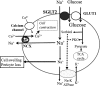
 ) Na+-K+ ATPase.
) Na+-K+ ATPase.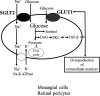
Similar articles
-
Roles of Sodium-Glucose Cotransporter 2 of Mesangial Cells in Diabetic Kidney Disease.J Endocr Soc. 2021 May 7;5(8):bvab083. doi: 10.1210/jendso/bvab083. eCollection 2021 Aug 1. J Endocr Soc. 2021. PMID: 34195526 Free PMC article. Review.
-
Lessons from the Trials for the Desirable Effects of Sodium Glucose Co-Transporter 2 Inhibitors on Diabetic Cardiovascular Events and Renal Dysfunction.Int J Mol Sci. 2019 Nov 12;20(22):5668. doi: 10.3390/ijms20225668. Int J Mol Sci. 2019. PMID: 31726765 Free PMC article. Review.
-
The presence of sodium glucose co-transporter 2 in mesangial cells and pericytes and its roles in mesangial lesions and in capillaries under diabetic and ischemic conditions.Diabetes Res Clin Pract. 2022 Oct;192:110096. doi: 10.1016/j.diabres.2022.110096. Epub 2022 Sep 27. Diabetes Res Clin Pract. 2022. PMID: 36174778
-
Sodium Glucose Cotransporter 2 (SGLT2) Plays as a Physiological Glucose Sensor and Regulates Cellular Contractility in Rat Mesangial Cells.PLoS One. 2016 Mar 21;11(3):e0151585. doi: 10.1371/journal.pone.0151585. eCollection 2016. PLoS One. 2016. PMID: 26999015 Free PMC article.
-
Sodium/glucose cotransporter 2 inhibitors and prevention of diabetic nephropathy: targeting the renal tubule in diabetes.Am J Kidney Dis. 2014 Jul;64(1):16-24. doi: 10.1053/j.ajkd.2014.02.010. Epub 2014 Mar 25. Am J Kidney Dis. 2014. PMID: 24673844 Review.
Cited by
-
Empagliflozin and the Risk of Retinopathy in Patients With Type 2 Diabetes.JAMA Ophthalmol. 2025 Jan 1;143(1):62-71. doi: 10.1001/jamaophthalmol.2024.5219. JAMA Ophthalmol. 2025. PMID: 39636645
-
Sodium-Glucose Cotransporter 2 Inhibitors and Risk of Retinopathy in Patients With Type 2 Diabetes.JAMA Netw Open. 2023 Dec 1;6(12):e2348431. doi: 10.1001/jamanetworkopen.2023.48431. JAMA Netw Open. 2023. PMID: 38117497 Free PMC article.
-
Sodium-glucose co-transporter 2 inhibitors and diabetic retinopathy: insights into preservation of sight and looking beyond.Cardiovasc Endocrinol Metab. 2020 May 18;10(1):3-13. doi: 10.1097/XCE.0000000000000209. eCollection 2021 Mar. Cardiovasc Endocrinol Metab. 2020. PMID: 33634250 Free PMC article. Review.
-
Recovery from Diabetic Macular Edema in a Diabetic Patient After Minimal Dose of a Sodium Glucose Co-Transporter 2 Inhibitor.Am J Case Rep. 2018 Apr 19;19:462-466. doi: 10.12659/ajcr.909708. Am J Case Rep. 2018. PMID: 29670074 Free PMC article.
-
Relationship between Diabetic Nephropathy and Development of Diabetic Macular Edema in Addition to Diabetic Retinopathy.Biomedicines. 2023 May 22;11(5):1502. doi: 10.3390/biomedicines11051502. Biomedicines. 2023. PMID: 37239172 Free PMC article.
References
-
- Armulik A, Abramsson A, Betsholtz C. 2005. Endothelial/pericyte interactions. Circ Res. 97:512–523. - PubMed
-
- Benfield P. 1986. Aldose reductase inhibitors and late complications of diabetes. Drugs. 32:43–55. - PubMed
-
- Bonner C, Kerr-Conte J, Gmyr V, Queniat G, Moerman E, Thévenet J, Beaucamps C, Delalleau N, Popescu I, Malaisse WJ. 2015. Inhibition of the glucose transporter SGLT2 with dapagliflozin in pancreatic alpha cells triggers glucagon secretion. Nat Med. 21:512–517. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

