Evaluation of Anthelmintic Resistance and Exhaust Air Dust PCR as a Diagnostic Tool in Mice Enzootically Infected with Aspiculuris tetraptera
- PMID: 28535863
- PMCID: PMC5438921
Evaluation of Anthelmintic Resistance and Exhaust Air Dust PCR as a Diagnostic Tool in Mice Enzootically Infected with Aspiculuris tetraptera
Abstract
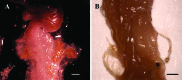
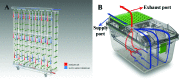
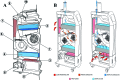
The entry of infectious agents in rodent colonies occurs despite robust sentinel monitoring programs, strict quarantine measures, and stringent biosecurity practices. In light of several outbreaks with Aspiculuris tetraptera in our facilities, we investigated the presence of anthelmintic resistance and the use of exhaust air dust (EAD) PCR for early detection of A. tetraptera infection. To determine anthelmintic resistance, C57BL/6, DBA/2, and NCr nude mice were experimentally inoculated with embryonated A. tetraptera ova harvested from enzootically infected mice, followed by treatment with 150 ppm fenbendazole in feed, 150 ppm fenbendazole plus 5 ppm piperazine in feed, or 2.1 mg/mL piperazine in water for 4 or 8 wk. Regardless of the mouse strain or treatment, no A. tetraptera were recovered at necropsy, indicating the lack of resistance in the worms to anthelmintic treatment. In addition, 10 of 12 DBA/2 positive-control mice cleared the A. tetraptera infection without treatment. To evaluate the feasibility of EAD PCR for A. tetraptera, 69 cages of breeder mice enzootically infected with A. tetraptera were housed on a Tecniplast IVC rack as a field study. On day 0, 56% to 58% of the cages on this rack tested positive for A. tetraptera by PCR and fecal centrifugation flotation (FCF). PCR from EAD swabs became positive for A. tetraptera DNA within 1 wk of placing the above cages on the rack. When these mice were treated with 150 ppm fenbendazole in feed, EAD PCR reverted to pinworm-negative after 1 mo of treatment and remained negative for an additional 8 wk. The ability of EAD PCR to detect few A. tetraptera positive mice was investigated by housing only 6 infected mice on another IVC rack as a field study. The EAD PCR from this rack was positive for A. tetraptera DNA within 1 wk of placing the positive mice on it. These findings demonstrate that fenbendazole is still an effective anthelmintic and that EAD PCR is a rapid, noninvasive assay that may be a useful diagnostic tool for antemortem detection of A. tetraptera infection, in conjunction with fecal PCR and FCF.
Figures






Similar articles
-
False-positive results after environmental pinworm PCR testing due to Rhabditid nematodes in Corncob bedding.J Am Assoc Lab Anim Sci. 2014 Nov;53(6):717-24. J Am Assoc Lab Anim Sci. 2014. PMID: 25650980 Free PMC article.
-
Evaluation of coinfection with pinworms (Aspiculuris tetraptera, Dentostomella translucida, and Syphacia obvelata) in gerbils and mice.Contemp Top Lab Anim Sci. 2003 Jul;42(4):46-8. Contemp Top Lab Anim Sci. 2003. PMID: 12906402
-
Comparison of traditional and PCR methods during screening for and confirmation of Aspiculuris tetraptera in a mouse facility.J Am Assoc Lab Anim Sci. 2011 Nov;50(6):904-9. J Am Assoc Lab Anim Sci. 2011. PMID: 22330785 Free PMC article.
-
Pinworm infections in laboratory rodents: a review.Lab Anim. 1976 Jan;10(1):1-13. doi: 10.1258/002367776780948862. Lab Anim. 1976. PMID: 768631 Review.
-
A review of treatments for the eradication of pinworm infections from laboratory rodent colonies.Contemp Top Lab Anim Sci. 2002 Mar;41(2):36-46. Contemp Top Lab Anim Sci. 2002. PMID: 11958602 Review.
Cited by
-
An Update on the Biologic Effects of Fenbendazole.Comp Med. 2022 Aug 1;72(4):215-219. doi: 10.30802/AALAS-CM-22-000006. Epub 2022 Jun 28. Comp Med. 2022. PMID: 35764389 Free PMC article.
-
Evaluation of In-cage Filter Paper as a Replacement for Sentinel Mice in the Detection of Murine Pathogens.J Am Assoc Lab Anim Sci. 2021 Mar 1;60(2):160-167. doi: 10.30802/AALAS-JAALAS-20-000086. Epub 2021 Feb 24. J Am Assoc Lab Anim Sci. 2021. PMID: 33629939 Free PMC article.
-
Adoption of Exhaust Air Dust Testing in SPF Rodent Facilities.J Am Assoc Lab Anim Sci. 2020 Mar 1;59(2):156-162. doi: 10.30802/AALAS-JAALAS-19-000079. Epub 2020 Feb 19. J Am Assoc Lab Anim Sci. 2020. PMID: 32075699 Free PMC article.
-
Comparison of Rodent Infectious Agent Detection by Exhaust Dust Testing and Traditional Sentinel Testing Using Quantitative Polymerase Chain Reaction.J Am Assoc Lab Anim Sci. 2024 May 7;63(5):530-9. doi: 10.30802/AALAS-JAALAS-23-000125. Online ahead of print. J Am Assoc Lab Anim Sci. 2024. PMID: 38714357 Free PMC article.
-
Health Monitoring of Laboratory Rodent Colonies-Talking about (R)evolution.Animals (Basel). 2021 May 14;11(5):1410. doi: 10.3390/ani11051410. Animals (Basel). 2021. PMID: 34069175 Free PMC article. Review.
References
-
- Anya AO. 1966. Experimental studies on the physiology of hatching of eggs of Aspiculuris tetraptera Schulz (Oxyuridea: Nematoda). Parasitology 56:733–744. - PubMed
-
- Anya AO. 1966. Studies on the biology of some oxyurid nematodes. I. Factors in the development of eggs of Aspiculuris tetraptera Schulz. J Helminthol 40:253–260. - PubMed
-
- Baird SM, Beattie GM, Lannom RA, Lipsick JS, Jensen FC, Kaplan NO. 1982. Induction of lymphoma in antigenically stimulated athymic mice. Cancer Res 42:198–206. - PubMed
-
- Barthold SW, Griffey SM, Percy DH. 2016. Mouse, p 1–117. In: Barthold SW, Griffey SM, Percy DH. Pathology of laboratory rodents and rabbits, 4th ed Ames (IA): Wiley Blackwell.
-
- Beattie GM, Baird SM, Lipsick JS, Lannom RA, Kaplan NO. 1981. Induction of T and B lymphocyte responses in antigenically stimulated athymic mice. Cancer Res 41:2322–2327. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
