Circulating Heparan Sulfate Fragments Attenuate Histone-Induced Lung Injury Independently of Histone Binding
- PMID: 28538085
- PMCID: PMC5685884
- DOI: 10.1097/SHK.0000000000000907
Circulating Heparan Sulfate Fragments Attenuate Histone-Induced Lung Injury Independently of Histone Binding
Abstract
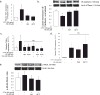
Extracellular histones are cationic damage-associated molecular pattern molecules capable of directly inducing cellular injury via charge-mediated interactions with plasma membranes. Accordingly, histones released into the plasma during critical illness are known to contribute to the onset and propagation of lung injury. Vascular injury (with consequent degradation of the endothelial glycocalyx) simultaneously releases anionic heparan sulfate fragments (hexa- to octasaccharides in size) into the plasma. It is unknown whether this endogenous release of heparan sulfate fragments modulates charge-dependent histone cytotoxicity, or if exogenous heparan sulfate fragments could therapeutically attenuate histone-induced lung injury. Using isothermic calorimetry, we found that extracellular histones only bind to heparan sulfate fragments ≥ 10 saccharides in size, suggesting that glycocalyx-derived heparan sulfate hexa/octasaccharides are incapable of intercepting/neutralizing circulating histones. However, we found that even heparan sulfate fragments incapable of histone binding (e.g., tetrasaccharides) attenuated histone-induced lung injury in vivo, suggesting a direct, size-independent protective effect of heparan sulfate. We found that histones had no effect on human neutrophils ex vivo but exerted toll-like receptor-independent cytotoxicity on human pulmonary microvascular endothelial cells in vitro. This cytotoxicity could be prevented by either the addition of negatively charged (i.e., highly sulfated) heparan sulfate tetrasaccharides (incapable of binding histones) or decasaccharides (capable of binding histones). Taken together, our findings suggest that heparan sulfate oligosaccharides may directly exert pulmonary endothelial-protective effects that attenuate histone-mediated lung injury.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



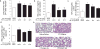
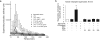
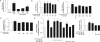
References
-
- Allam R, Kumar SV, Darisipudi MN, Anders HJ. Extracellular histones in tissue injury and inflammation. J Mol Med (Berl) 2014;92(5):465–72. - PubMed
-
- Freeman CG, Parish CR, Knox KJ, Blackmore JL, Lobov SA, King DW, Senden TJ, Stephens RW. The accumulation of circulating histones on heparan sulphate in the capillary glycocalyx of the lungs. Biomaterials. 2013;34(22):5670–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

