miR-29b contributes to multiple types of muscle atrophy
- PMID: 28541289
- PMCID: PMC5458521
- DOI: 10.1038/ncomms15201
miR-29b contributes to multiple types of muscle atrophy
Abstract
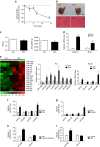
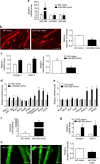
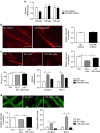
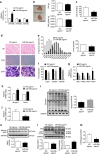
A number of microRNAs (miRNAs, miRs) have been shown to play a role in skeletal muscle atrophy, but their role is not completely understood. Here we show that miR-29b promotes skeletal muscle atrophy in response to different atrophic stimuli in cells and in mouse models. miR-29b promotes atrophy of myotubes differentiated from C2C12 or primary myoblasts, and conversely, its inhibition attenuates atrophy induced by dexamethasone (Dex), TNF-α and H2O2 treatment. Targeting of IGF-1 and PI3K(p85α) by miR-29b is required for induction of muscle atrophy. In vivo, miR-29b overexpression is sufficient to promote muscle atrophy while inhibition of miR-29b attenuates atrophy induced by denervation and immobilization. These data suggest that miR-29b contributes to multiple types of muscle atrophy via targeting of IGF-1 and PI3K(p85α), and that suppression of miR-29b may represent a therapeutic approach for muscle atrophy induced by different stimuli.
Conflict of interest statement
The authors declare no competing financial interests.
Figures




Similar articles
-
MicroRNA 322 Aggravates Dexamethasone-Induced Muscle Atrophy by Targeting IGF1R and INSR.Int J Mol Sci. 2020 Feb 7;21(3):1111. doi: 10.3390/ijms21031111. Int J Mol Sci. 2020. PMID: 32046161 Free PMC article.
-
miR-182 attenuates atrophy-related gene expression by targeting FoxO3 in skeletal muscle.Am J Physiol Cell Physiol. 2014 Aug 15;307(4):C314-9. doi: 10.1152/ajpcell.00395.2013. Epub 2014 May 28. Am J Physiol Cell Physiol. 2014. PMID: 24871856 Free PMC article.
-
miR-23a is decreased during muscle atrophy by a mechanism that includes calcineurin signaling and exosome-mediated export.Am J Physiol Cell Physiol. 2014 Mar 15;306(6):C551-8. doi: 10.1152/ajpcell.00266.2013. Epub 2013 Dec 11. Am J Physiol Cell Physiol. 2014. PMID: 24336651 Free PMC article.
-
MicroRNA in myogenesis and muscle atrophy.Curr Opin Clin Nutr Metab Care. 2013 May;16(3):258-66. doi: 10.1097/MCO.0b013e32835f81b9. Curr Opin Clin Nutr Metab Care. 2013. PMID: 23449000 Free PMC article. Review.
-
Regulation of muscle atrophy by microRNAs: 'AtromiRs' as potential target in cachexia.Curr Opin Clin Nutr Metab Care. 2018 Nov;21(6):423-429. doi: 10.1097/MCO.0000000000000503. Curr Opin Clin Nutr Metab Care. 2018. PMID: 30198917 Review.
Cited by
-
MicroRNAs and their Modulatory Effect on the Hallmarks of Osteosarcopenia.Curr Osteoporos Rep. 2024 Oct;22(5):458-470. doi: 10.1007/s11914-024-00880-4. Epub 2024 Aug 20. Curr Osteoporos Rep. 2024. PMID: 39162945 Review.
-
Botulinum toxin A-induced muscle paralysis stimulates Hdac4 and differential miRNA expression.PLoS One. 2018 Nov 14;13(11):e0207354. doi: 10.1371/journal.pone.0207354. eCollection 2018. PLoS One. 2018. PMID: 30427927 Free PMC article.
-
miR-27b-3p Attenuates Muscle Atrophy by Targeting Cbl-b in Skeletal Muscles.Biomolecules. 2022 Jan 23;12(2):191. doi: 10.3390/biom12020191. Biomolecules. 2022. PMID: 35204692 Free PMC article.
-
Tetrandrine Inhibits Skeletal Muscle Differentiation by Blocking Autophagic Flux.Int J Mol Sci. 2022 Jul 24;23(15):8148. doi: 10.3390/ijms23158148. Int J Mol Sci. 2022. PMID: 35897724 Free PMC article.
-
Dicer-mediated miRNA processing is not involved in controlling muscle mass during muscle atrophy.Sci Rep. 2021 Sep 29;11(1):19361. doi: 10.1038/s41598-021-98545-0. Sci Rep. 2021. PMID: 34588544 Free PMC article.
References
-
- Bodine S. C. et al.. Identification of ubiquitin ligases required for skeletal muscle atrophy. Science 294, 1704–1708 (2001). - PubMed
-
- Cohen S., Nathan J. A. & Goldberg A. L. Muscle wasting in disease: molecular mechanisms and promising therapies. Nat. Rev. Drug. Discov. 14, 58–74 (2015). - PubMed
-
- Dutt V., Gupta S., Dabur R., Injeti E. & Mittal A. Skeletal muscle atrophy: potential therapeutic agents and their mechanisms of action. Pharmacol. Res. 99, 86–100 (2015). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

