An inactivated hand-foot-and-mouth disease vaccine using the enterovirus 71 (C4a) strain isolated from a Korean patient induces a strong immunogenic response in mice
- PMID: 28542556
- PMCID: PMC5443535
- DOI: 10.1371/journal.pone.0178259
An inactivated hand-foot-and-mouth disease vaccine using the enterovirus 71 (C4a) strain isolated from a Korean patient induces a strong immunogenic response in mice
Abstract
Enterovirus 71 (EV71) is a major causative agent of hand-foot-and-mouth disease (HFMD) frequently occurring in children. HFMD induced by EV71 can cause serious health problems and has been reported worldwide, particularly in the Asia-Pacific region. In this study, we assessed the immunogenicity of a formalin-inactivated HFMD vaccine using an EV71 strain (FI-EV71 C4a) isolated from a Korean patient. The vaccine candidate was evaluated in mice to determine the vaccination doses and vaccine schedules. BALB/c mice were intramuscularly administered 5, 10, or 20 μg FI-EV71 vaccine, followed by a booster 2 weeks later. EV71-specific antibodies and neutralizing antibodies were induced and maintained until the end of the experimental period in all vaccinated groups. To determine the effectiveness of adjuvant for the EV71 vaccine, three adjuvants, i.e., aluminium hydroxide gel, monophosphoryl lipid A, and polyinosinic-polycytidylic acid, were administered separately with the FI-EV71 vaccine to mice via the intramuscular route. Mice administered the FI-EV71 vaccine formulated with all three adjuvants induced a significantly increased antibody response compared with that of the single adjuvant groups. The vaccinated group with triple adjuvants exhibited more rapid induction of EV71-specific and neutralizing antibodies than the other groups. These results suggested that the role of adjuvant in inactivated vaccine was important for eliciting effective immune responses against EV71. In conclusion, our results showed that FI-EV71 was a potential candidate vaccine for prevention of EV71 infection.
Conflict of interest statement
Figures
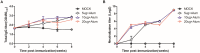
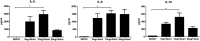
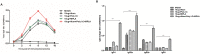

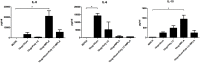
Similar articles
-
Enhanced neutralizing antibody response induced by inactivated enterovirus 71 in cynomolgus monkeys.PLoS One. 2018 Oct 2;13(10):e0202552. doi: 10.1371/journal.pone.0202552. eCollection 2018. PLoS One. 2018. PMID: 30278051 Free PMC article.
-
Inactivated enterovirus 71 with poly-γ-glutamic acid/Chitosan nano particles (PC NPs) induces high cellular and humoral immune responses in BALB/c mice.Arch Virol. 2018 Aug;163(8):2073-2083. doi: 10.1007/s00705-018-3837-3. Epub 2018 Apr 4. Arch Virol. 2018. PMID: 29619599
-
Production of EV71 vaccine candidates.Hum Vaccin Immunother. 2012 Dec 1;8(12):1775-83. doi: 10.4161/hv.21739. Epub 2012 Sep 19. Hum Vaccin Immunother. 2012. PMID: 22992566 Free PMC article. Review.
-
Evaluation of monovalent and bivalent vaccines against lethal Enterovirus 71 and Coxsackievirus A16 infection in newborn mice.Hum Vaccin Immunother. 2014;10(10):2885-95. doi: 10.4161/hv.29823. Hum Vaccin Immunother. 2014. PMID: 25483672 Free PMC article.
-
Review of enterovirus 71 vaccines.Clin Infect Dis. 2015 Mar 1;60(5):797-803. doi: 10.1093/cid/ciu852. Epub 2014 Oct 28. Clin Infect Dis. 2015. PMID: 25352588 Review.
Cited by
-
Enhanced neutralizing antibody response induced by inactivated enterovirus 71 in cynomolgus monkeys.PLoS One. 2018 Oct 2;13(10):e0202552. doi: 10.1371/journal.pone.0202552. eCollection 2018. PLoS One. 2018. PMID: 30278051 Free PMC article.
-
Enterovirus A71 Vaccines.Vaccines (Basel). 2021 Feb 27;9(3):199. doi: 10.3390/vaccines9030199. Vaccines (Basel). 2021. PMID: 33673595 Free PMC article. Review.
-
Seroprevalence of antibodies to enterovirus 71 and coxsackievirus A16 among people of various age groups in a northeast province of Thailand.Virol J. 2018 Oct 16;15(1):158. doi: 10.1186/s12985-018-1074-8. Virol J. 2018. PMID: 30326914 Free PMC article.
-
Hand, Foot and Mouth Disease in West Bengal, India: A Preliminary Report on Clinicovirological Trend over 3 Successive Years (2013-2015).Indian J Dermatol. 2017 Sep-Oct;62(5):486-490. doi: 10.4103/ijd.IJD_381_17. Indian J Dermatol. 2017. PMID: 28979010 Free PMC article.
-
A comprehensive overview of vaccines developed for pandemic viral pathogens over the past two decades including those in clinical trials for the current novel SARS-CoV-2.RSC Adv. 2021 Jun 8;11(33):20006-20035. doi: 10.1039/d0ra09668g. eCollection 2021 Jun 3. RSC Adv. 2021. PMID: 35479882 Free PMC article. Review.
References
-
- Wong SS, Yip CC, Lau SK, Yuen KY. Human enterovirus 71 and hand, foot and mouth disease. Epidemiology and infection. 2010;138(8):1071–89. Epub 2010/01/09. doi: 10.1017/S0950268809991555 - DOI - PubMed
-
- Cardosa MJ, Perera D, Brown BA, Cheon D, Chan HM, Chan KP, et al. Molecular epidemiology of human enterovirus 71 strains and recent outbreaks in the Asia-Pacific region: comparative analysis of the VP1 and VP4 genes. Emerging infectious diseases. 2003;9(4):461–8. Epub 2003/04/19. PubMed Central PMCID: PMCPMC2957976. doi: 10.3201/eid0904.020395 - DOI - PMC - PubMed
-
- McMinn P, Lindsay K, Perera D, Chan HM, Chan KP, Cardosa MJ. Phylogenetic analysis of enterovirus 71 strains isolated during linked epidemics in Malaysia, Singapore, and Western Australia. J Virol. 2001;75(16):7732–8. Epub 2001/07/20. PubMed Central PMCID: PMCPMC115010. doi: 10.1128/JVI.75.16.7732-7738.2001 - DOI - PMC - PubMed
-
- Lee M-S, Tseng F-C, Wang J-R, Chi C-Y, Chong P, Su I-J. Challenges to Licensure of Enterovirus 71 Vaccines. PLoS Negl Trop Dis. 2012;6(8):e1737 doi: 10.1371/journal.pntd.0001737 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

