Comparative lytic efficacy of rt-PA and ultrasound in porcine versus human clots
- PMID: 28545055
- PMCID: PMC5435301
- DOI: 10.1371/journal.pone.0177786
Comparative lytic efficacy of rt-PA and ultrasound in porcine versus human clots
Abstract
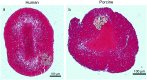
Introduction: Porcine thrombi are employed routinely in preclinical models of ischemic stroke. In this study, we examined the differential lytic susceptibility of porcine and human whole blood clots with and without the use of microbubbles and ultrasound (US) as an adjuvant.
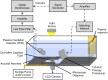
Materials and methods: An in vitro system equipped with time-lapse microscopy was used to evaluate recombinant tissue-plasminogen activator (rt-PA) lysis of porcine and human clots in the same species or cross species plasma. Human and porcine whole blood clots were treated with rt-PA and an echo contrast agent, Definity®, and exposed to intermittent 120 kHz US.
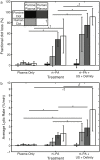
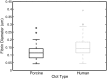
Results and conclusions: The rt-PA lytic efficacy observed for porcine clots in porcine plasma was 22 times lower than for human clots in human plasma reported previously. Further, porcine clots did not exhibit increased lysis with adjuvant Definity® and US exposure. However, the rt-PA lytic susceptibility of the porcine clots in human plasma was similar to that of human clots in human plasma. Human clots perfused with porcine plasma did not respond to rt-PA, but adjuvant use of Definity® and US enhanced lysis. These results reveal considerable differences in lytic susceptibility of porcine clots and human clots to rt-PA. The use of porcine clot models to test new human thrombolytic therapies may necessitate modulation of coagulation and thrombolytic factors to reflect human hemostasis accurately.
Conflict of interest statement
Figures


Similar articles
-
In vitro characterization of sonothrombolysis and echocontrast agents to treat ischemic stroke.Sci Rep. 2019 Jul 9;9(1):9902. doi: 10.1038/s41598-019-46112-z. Sci Rep. 2019. PMID: 31289285 Free PMC article.
-
Effect of Clot Stiffness on Recombinant Tissue Plasminogen Activator Lytic Susceptibility in Vitro.Ultrasound Med Biol. 2018 Dec;44(12):2710-2727. doi: 10.1016/j.ultrasmedbio.2018.08.005. Epub 2018 Sep 26. Ultrasound Med Biol. 2018. PMID: 30268531 Free PMC article.
-
Ultrasound-enhanced rt-PA thrombolysis in an ex vivo porcine carotid artery model.Ultrasound Med Biol. 2011 Aug;37(8):1240-51. doi: 10.1016/j.ultrasmedbio.2011.05.011. Ultrasound Med Biol. 2011. PMID: 21723448 Free PMC article.
-
Effect of low frequency ultrasound on combined rt-PA and eptifibatide thrombolysis in human clots.Thromb Res. 2009;123(3):528-36. doi: 10.1016/j.thromres.2008.05.011. Epub 2008 Jul 10. Thromb Res. 2009. PMID: 18619651 Free PMC article.
-
Direct microscopic monitoring of initial and dynamic clot lysis using plasmin or rt-PA in an in vitro flow system.Thromb Res. 2014 May;133(5):908-13. doi: 10.1016/j.thromres.2014.02.008. Epub 2014 Feb 22. Thromb Res. 2014. PMID: 24613694
Cited by
-
In vitro characterization of sonothrombolysis and echocontrast agents to treat ischemic stroke.Sci Rep. 2019 Jul 9;9(1):9902. doi: 10.1038/s41598-019-46112-z. Sci Rep. 2019. PMID: 31289285 Free PMC article.
-
Assessment of histological characteristics, imaging markers, and rt-PA susceptibility of ex vivo venous thrombi.Sci Rep. 2021 Nov 23;11(1):22805. doi: 10.1038/s41598-021-02030-7. Sci Rep. 2021. PMID: 34815441 Free PMC article.
-
Enhancing Passive Cavitation Imaging Using pth Root Compression Delay, Sum, and Integrate Beamforming: In Vitro and In Vivo Studies.IEEE Trans Biomed Eng. 2025 Jul;72(7):2283-2292. doi: 10.1109/TBME.2025.3540101. IEEE Trans Biomed Eng. 2025. PMID: 40031858
-
Effect of Thrombin and Incubation Time on Porcine Whole Blood Clot Elasticity and Recombinant Tissue Plasminogen Activator Susceptibility.Ultrasound Med Biol. 2022 Aug;48(8):1567-1578. doi: 10.1016/j.ultrasmedbio.2022.04.003. Epub 2022 May 26. Ultrasound Med Biol. 2022. PMID: 35644763 Free PMC article.
-
Accelerated sonothrombolysis with Definity in a xenographic porcine cerebral thromboembolism model.Sci Rep. 2021 Feb 17;11(1):3987. doi: 10.1038/s41598-021-83442-3. Sci Rep. 2021. PMID: 33597659 Free PMC article.
References
-
- Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, et al. Heart Disease and Stroke Statistics—2016 Update. Circulation [Internet]. 2016. January 26;133(4):e38–360. Available from: http://circ.ahajournals.org/lookup/doi/10.1161/CIR.0000000000000350 - DOI - PubMed
-
- The NINDS rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med [Internet]. 1995. December 14 [cited 2013 Nov 10];333(24):1581–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/7477192 doi: 10.1056/NEJM199512143332401 - DOI - PubMed
-
- Hacke W, Kaste M, Bluhmki E, Brozman M, Dávalos A, Guidetti D, et al. Thrombolysis with Alteplase 3 to 4.5 Hours after Acute Ischemic Stroke. N Engl J Med [Internet]. 2008. September 25;359(13):1317–29. Available from: http://www.nejm.org/doi/abs/10.1056/NEJMoa0804656 - DOI - PubMed
-
- Zivin JA, Fisher M, DeGirolami U, Hemenway CC, Stashak JA. Tissue plasminogen activator reduces neurological damage after cerebral embolism. Science. 1985;230(4731):1289–92. - PubMed
-
- Suarez JI, Sunshine JL, Tarr R, Zaidat O, Selman WR, Kernich C, et al. Predictors of Clinical Improvement, Angiographic Recanalization, and Intracranial Hemorrhage After Intra-Arterial Thrombolysis for Acute Ischemic Stroke. Stroke [Internet]. 1999. October 1;30(10):2094–100. Available from: http://stroke.ahajournals.org/cgi/doi/10.1161/01.STR.30.10.2094 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

