Transmission Electron Microscopy as a Tool for the Characterization of Soft Materials: Application and Interpretation
- PMID: 28546914
- PMCID: PMC5441488
- DOI: 10.1002/advs.201600476
Transmission Electron Microscopy as a Tool for the Characterization of Soft Materials: Application and Interpretation
Abstract
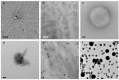
Transmission electron microscopy (TEM) provides direct structural information on nano-structured materials and is popular as a characterization tool in soft matter and supramolecular chemistry. However, technical aspects of sample preparation are overlooked and erroneous image interpretations are regularly encountered in the literature. There are three most commonly used TEM methods as we derived from literature: drying, staining and cryo-TEM, which are explained here with respect to their application, limitations and interpretation. Since soft matter chemistry relies on a lot of indirect evidence, the role of TEM for the correct evaluation of the nature of an assembly is very large. Mistakes in application and interpretation can therefore have enormous impact on the quality of present and future studies. We provide helpful background information of these three techniques, the information that can and cannot be derived from them and provide assistance in selecting the right technique for soft matter imaging. This essay warns against the use of drying and explains why. In general cryo-TEM is by far the best suited method and many mistakes and over-interpretations can be avoided by the use of this technique.
Keywords: artefacts; cryo‐TEM; sample preparation; self‐assembly; vesicles.
Figures




Similar articles
-
Imaging of self-assembled structures: interpretation of TEM and cryo-TEM images.Angew Chem Int Ed Engl. 2010 Oct 18;49(43):7850-8. doi: 10.1002/anie.201001493. Angew Chem Int Ed Engl. 2010. PMID: 20821772
-
Characterizing the Core-Shell Architecture of Block Copolymer Nanoparticles with Electron Microscopy: A Multi-Technique Approach.Polymers (Basel). 2020 Jul 25;12(8):1656. doi: 10.3390/polym12081656. Polymers (Basel). 2020. PMID: 32722462 Free PMC article.
-
Progress in the direct structural characterization of fibrous amphiphilic supramolecular assemblies in solution by transmission electron microscopic techniques.Adv Colloid Interface Sci. 2014 Jun;208:279-92. doi: 10.1016/j.cis.2014.01.007. Epub 2014 Jan 24. Adv Colloid Interface Sci. 2014. PMID: 24508499 Review.
-
Elucidating the assembled structure of amphiphiles in solution via cryogenic transmission electron microscopy.Soft Matter. 2007 Jul 17;3(8):945-955. doi: 10.1039/b704194b. Soft Matter. 2007. PMID: 32900043
-
Cryo-FIB for TEM investigation of soft matter and beam sensitive energy materials.Nanotechnology. 2022 Oct 4;33(50). doi: 10.1088/1361-6528/ac92eb. Nanotechnology. 2022. PMID: 36121746 Review.
Cited by
-
Peg-Grafted Liposomes for L-Asparaginase Encapsulation.Pharmaceutics. 2022 Aug 29;14(9):1819. doi: 10.3390/pharmaceutics14091819. Pharmaceutics. 2022. PMID: 36145567 Free PMC article.
-
Computational Reverse Engineering Analysis for Scattering Experiments (CREASE) on Vesicles Assembled from Amphiphilic Macromolecular Solutions.JACS Au. 2021 Sep 29;1(11):1925-1936. doi: 10.1021/jacsau.1c00305. eCollection 2021 Nov 22. JACS Au. 2021. PMID: 34841410 Free PMC article.
-
Cargo shuttling by electrochemical switching of core-shell microgels obtained by a facile one-shot polymerization.Chem Sci. 2018 Dec 13;10(6):1844-1856. doi: 10.1039/c8sc04369h. eCollection 2019 Feb 14. Chem Sci. 2018. PMID: 30842853 Free PMC article.
-
Correct Identification of the Core-Shell Structure of Cell Membrane-Coated Polymeric Nanoparticles.Chemistry. 2022 Dec 6;28(68):e202200947. doi: 10.1002/chem.202200947. Epub 2022 Oct 19. Chemistry. 2022. PMID: 36116117 Free PMC article.
-
Enhancing Ezetimibe Anticancer Activity Through Development of Drug Nano-Micelles Formulations: A Promising Strategy Supported by Molecular Docking.Int J Nanomedicine. 2023 Nov 15;18:6689-6703. doi: 10.2147/IJN.S438704. eCollection 2023. Int J Nanomedicine. 2023. PMID: 38026536 Free PMC article.
References
-
- Lehn J. M., Angew. Chem. Int. Ed. 1990, 29, 1304.
-
- Corbett P. T., Leclaire J., Vial L., West K. R., Wietor J., Sanders J. K. M., Otto S., Chem. Rev. 2006, 106, 3652. - PubMed
-
- Ludlow R. F., Otto S., Chem. Soc. Rev. 2008, 37, 101. - PubMed
-
- Coleman A. C., Beierle J. M., Stuart M. C. A., Macia B., Caroli G., Mika J. T., van Dijken D. J., Chen J., Browne W. R., Feringa B. L., Nat. Nanotechnol. 2011, 6, 547. - PubMed
-
- Holder S. J., Sommerdijk N. A. J. M., Polym. Chem. 2011, 2, 1018.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
