Plumbagin suppresses chronic periodontitis in rats via down-regulation of TNF-α, IL-1β and IL-6 expression
- PMID: 28552911
- PMCID: PMC5547554
- DOI: 10.1038/aps.2017.19
Plumbagin suppresses chronic periodontitis in rats via down-regulation of TNF-α, IL-1β and IL-6 expression
Abstract
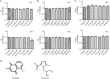
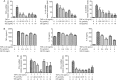
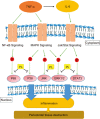
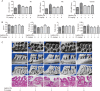
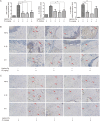
Chronic periodontitis (CP) is one of the most common oral diseases, which causes alveolar bone absorption and tooth loss in adults. In this study we aimed to investigate the potential of plumbagin (PL), a widely-investigated active compound extracted from the traditional Chinese herb Plumbago zeylanica L in treating CP. Human periodontal ligament stem cells (PDLSCs) were used for in vitro studies, whereas an animal model of CP was established in SD rats by ligation+Porphyromonas gingivalis (Pg) stimulation. The rats were injected with PL (2, 4, and 6 mg·kg-1·d-1, ip) for 4 weeks. Treatment of PDLSCs with TNF-α (10 ng/mL) markedly stimulated the expression of the proinflammatory cytokines TNF-α, IL-1β and IL-6, as well as the chemokines CCL-2 and CCL-5, which were dose-dependently suppressed by co-treatment with PL (1.25-5 μmol/L). Furthermore, PL (3.75 μmol/L) markedly suppressed TNF-α-induced activation of the MAPK, NF-κB and JAK/STAT signaling pathways in PDLSCs. In consistence with the in vitro studies, PL administration significantly decreased the expression of TNF-α, IL-1β and IL-6 in gingiva of the rat with CP, with the dosage 4 mg·kg-1·d-1 showing the best anti-inflammatory effect. Moreover, PL administration decelerated bone destruction in the rat with CP, evidenced by the aveolar bone loss (ABL) and H&E staining results. In conclusion, PL suppresses CP progression in rats by downregulating the expressions of TNF-α, IL-1β and IL-6 and inhibiting the MAPK, NF-κB and JAK/STAT signaling pathways.
Figures


Similar articles
-
Plumbagin inhibits LPS-induced inflammation through the inactivation of the nuclear factor-kappa B and mitogen activated protein kinase signaling pathways in RAW 264.7 cells.Food Chem Toxicol. 2014 Feb;64:177-83. doi: 10.1016/j.fct.2013.11.027. Epub 2013 Dec 1. Food Chem Toxicol. 2014. PMID: 24296134
-
The Proteasome Inhibitor Bortezomib Inhibits Inflammatory Response of Periodontal Ligament Cells and Ameliorates Experimental Periodontitis in Rats.J Periodontol. 2017 May;88(5):473-483. doi: 10.1902/jop.2016.160396. Epub 2016 Dec 16. J Periodontol. 2017. PMID: 27982724
-
Preventive effects of the novel antimicrobial peptide Nal-P-113 in a rat Periodontitis model by limiting the growth of Porphyromonas gingivalis and modulating IL-1β and TNF-α production.BMC Complement Altern Med. 2017 Aug 29;17(1):426. doi: 10.1186/s12906-017-1931-9. BMC Complement Altern Med. 2017. PMID: 28851350 Free PMC article.
-
Comparison of CCL28, interleukin-8, interleukin-1β and tumor necrosis factor-alpha in subjects with gingivitis, chronic periodontitis and generalized aggressive periodontitis.J Periodontal Res. 2013 Feb;48(1):44-51. doi: 10.1111/j.1600-0765.2012.01500.x. Epub 2012 Jul 19. J Periodontal Res. 2013. PMID: 22812409
-
Chronic Inflammation as a Link between Periodontitis and Carcinogenesis.Mediators Inflamm. 2019 Mar 27;2019:1029857. doi: 10.1155/2019/1029857. eCollection 2019. Mediators Inflamm. 2019. PMID: 31049022 Free PMC article. Review.
Cited by
-
Treatment with Luteolin Improves Lipopolysaccharide-Induced Periodontal Diseases in Rats.Biomedicines. 2020 Oct 21;8(10):442. doi: 10.3390/biomedicines8100442. Biomedicines. 2020. PMID: 33096800 Free PMC article.
-
Effects of sitagliptin activation of the stromal cell-derived factor-1/CXC chemokine receptor 4 signaling pathway on the proliferation, apoptosis, inflammation, and osteogenic differentiation of human periodontal ligament stem cells induced by lipopolysaccharide.Hua Xi Kou Qiang Yi Xue Za Zhi. 2024 Feb 1;42(1):37-45. doi: 10.7518/hxkq.2024.2023213. Hua Xi Kou Qiang Yi Xue Za Zhi. 2024. PMID: 38475949 Free PMC article. Chinese, English.
-
New insights into nanotherapeutics for periodontitis: a triple concerto of antimicrobial activity, immunomodulation and periodontium regeneration.J Nanobiotechnology. 2024 Jan 4;22(1):19. doi: 10.1186/s12951-023-02261-y. J Nanobiotechnology. 2024. PMID: 38178140 Free PMC article. Review.
-
Plumbagin alleviates temporomandibular joint osteoarthritis progression by inhibiting chondrocyte ferroptosis via the MAPK signaling pathways.Aging (Albany NY). 2023 Nov 29;15(22):13452-13470. doi: 10.18632/aging.205253. Epub 2023 Nov 29. Aging (Albany NY). 2023. PMID: 38032278 Free PMC article.
-
Lactobacillus helveticus Prevents Periodontitis Induced by Aggregatibacter actinomycetemcomitans in Rats by Regulating β-Defensins.Comput Math Methods Med. 2022 Feb 27;2022:4968016. doi: 10.1155/2022/4968016. eCollection 2022. Comput Math Methods Med. 2022. Retraction in: Comput Math Methods Med. 2023 Sep 27;2023:9816032. doi: 10.1155/2023/9816032. PMID: 35265172 Free PMC article. Retracted.
References
-
- Flemmig TF. Periodontitis. Ann Periodontol 1999; 4: 32–8. - PubMed
-
- Amano A. Bacterial adhesins to host components in periodontitis. Periodontol 2000 2010; 52: 12–37. - PubMed
-
- Feres M, Figueiredo LC, Soares GM, Faveri M. Systemic antibiotics in the treatment of periodontitis. Periodontol 2000 2015; 67: 131–86. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

