Epidural analgesia in critically ill patients with acute pancreatitis: the multicentre randomised controlled EPIPAN study protocol
- PMID: 28554928
- PMCID: PMC5730003
- DOI: 10.1136/bmjopen-2016-015280
Epidural analgesia in critically ill patients with acute pancreatitis: the multicentre randomised controlled EPIPAN study protocol
Erratum in
-
Correction: Epidural analgesia in critically ill patients with acute pancreatitis: the multicentre randomised controlled EPIPAN study protocol.BMJ Open. 2018 Feb 10;8(2):e015280corr1. doi: 10.1136/bmjopen-2016-015280corr1. BMJ Open. 2018. PMID: 29440166 Free PMC article. No abstract available.
Abstract
Background: Acute pancreatitis (AP) is associated with high morbidity and mortality in its most severe forms. Most patients with severe AP require intubation and invasive mechanical ventilation, frequently for more than 7 days, which is associated with the worst outcome. Recent increasing evidence from preclinical and clinical studies support the beneficial effects of epidural analgesia (EA) in AP, such as increased gut barrier function and splanchnic, pancreatic and renal perfusion, decreased liver damage and inflammatory response, and reduced mortality. Because recent studies suggest that EA might be a safe procedure in the critically ill, we sought to determine whether EA reduced AP-associated respiratory failure and other major clinical outcomes in patients with AP.
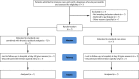
Methods and analysis: The Epidural Analgesia for Pancreatitis (EPIPAN) trial is an investigator-initiated, prospective, multicentre, randomised controlled two-arm trial with assessor-blinded outcome assessment. The EPIPAN trial will randomise 148 patients with AP requiring admission to an intensive care unit (ICU) to receive EA (with patient-controlled epidural administration of ropivacaine and sufentanil) combined with standard care based on current recommendations on the treatment of AP (interventional group), or standard care alone (reference group). The primary outcome is the number of ventilator-free days at day 30. Secondary outcomes include main complications of AP (eg, organ failure and mortality, among others), levels of biological markers of systemic inflammation, epithelial lung injury, renal failure, and healthcare-associated costs.
Ethics and dissemination: The study was approved by the appropriate ethics committee (CPP Sud-Est VI). Informed consent is required. If the combined application of EA and standard care proves superior to standard care alone in patients with AP in the ICU, the use of EA may become standard practice in experienced centres, thereby decreasing potential complications related to AP and its burden in critically ill patients. The results will be disseminated in a peer-reviewed journal.
Trial registration number: NCT02126332.
Keywords: Acute pancreatitis; Epidural analgesia; Intensive care unit; Randomised controlled trial.
© Article author(s) (or their employer(s) unless otherwise stated in the text of the article) 2017. All rights reserved. No commercial use is permitted unless otherwise expressly granted.
Conflict of interest statement
Competing interests: None declared.
Figures
References
-
- Acute respiratory distress syndrome: the Berlin definition. JAMA 2012;307. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials